Research Progress on Influencing Factors and Mechanisms of Chromium Valence State Transformation in Soil
-
摘要:
土壤中铬(Cr)污染是全球性环境问题,六价铬Cr(Ⅵ)因其高毒性及致癌性而成为关注焦点。土壤中的Cr主要以Cr(Ⅲ)和Cr(Ⅵ) )的形式存在,两者之间的转化受到诸如土壤pH值、氧化还原电位(Eh)、天然氧化还原剂、有机质和微生物的影响。随着工农业活动的增多,土壤中的Cr浓度不断增加,并通过植物吸收进入食物链等多种途径,对生态系统和人体健康构成威胁。本文对Cr在全球范围内的污染现状及来源、土壤中Cr的不同价态及其毒性特征进行了评述,并分析了pH、Eh对土壤中Cr的具体存在形态与价态的影响,及天然氧化还原剂、有机质等因素利用自身化学性质充当电子供体或受体的角色对土壤中Cr价态进行转化的氧化还原机理,以及不同影响因素之间的相互作用关系,从而可以全面理解土壤中Cr价态转化的行为机制。此外,在对Cr价态影响因素深入了解的基础上,总结了以生物炭和纳米材料为代表的先进修复技术方法,这些材料和方法由于能够将Cr(Ⅵ) 有效地还原为毒性较小的Cr(Ⅲ),从而降低生态环境风险,因而是一类具有巨大潜在应用价值的修复材料和修复方法,但大规模应用的可行性及其修复效果仍需要进一步验证。
要点(1) 土壤中的Cr(Ⅵ)主要以铬酸盐(CrO4 2−)、氢铬酸盐(HCrO4 − )和重铬酸盐(Cr2O7 2−)等氧化离子的形式存在,是土壤中毒性最大的Cr价态。
(2) 土壤中Cr价态受Eh、有机质等多种因素影响,这些因素可以促进或抑制Cr(Ⅵ)的还原。
(3) 生物炭和纳米材料等新型修复技术能够将Cr(Ⅵ)有效地还原为低毒性的Cr(Ⅲ),是治理Cr(Ⅵ)污染的一种可行技术方案。
HIGHLIGHTS(1) Cr(Ⅵ) in soil mainly exists in the form of oxidized ions such as chromate (CrO4 2− ), hydrogen chromate (HCrO4 − ), and dichromate (Cr2O7 2− ), and is the most toxic Cr valence state in soil.
(2) The valence state of Cr in soil is affected by Eh, organic matter and other factors, which can promote or inhibit the reduction of Cr(Ⅵ).
(3) New remediation technologies such as biochar and nanomaterials can effectively reduce Cr(Ⅵ) to low toxicity Cr(Ⅲ), which is a feasible technical solution for treating Cr(Ⅵ) pollution.
Abstract:Chromium (Cr) pollution in soil is a global environmental problem, and hexavalent chromium [Cr(Ⅵ)] has become a focus of attention due to its high toxicity and carcinogenicity. Cr in soil mainly exists in the form of Cr(Ⅲ) and Cr(Ⅵ), and the transformation between the two is influenced by factors such as soil pH, redox potential (Eh), natural redox agents, organic matter, and microorganisms. This article provides an overview of the global pollution status and sources of Cr in soil, as well as the different valence states and toxicity characteristics of Cr in soil. It also analyzes the redox mechanisms of chromium valence state transformation in soil affected by different factors, as well as the interactions between different factors. In addition, through a deep understanding of the factors affecting the valence state of Cr, advanced remediation techniques represented by biochar and nanomaterials have emerged. These methods can effectively reduce Cr(Ⅵ) to less toxic Cr(Ⅲ), thereby reducing ecological and environmental risks. Therefore, they are a potentially valuable remediation material and technique. However, the feasibility and effectiveness of large-scale applications still need further verification. The BRIEF REPORT is available for this paper at http://www.ykcs.ac.cn/en/article/doi/10.15898/j.ykcs.202401180007.
-
Keywords:
- chromium pollution /
- chromium valence state /
- natural redox agent /
- organic matter; remediation technology
This page contains the following errors:
error on line 1 at column 32: Entity 'nbsp' not definedBelow is a rendering of the page up to the first error.
-
土壤矿物中能够被作物直接吸收利用的部分,即有效量,以活动态存在于土壤中[1-2]。元素对环境或生物体产生的效应或毒性,在很大程度上取决于其各形态含量,而不是总浓度[3-4]。土壤微量元素有效量不但影响作物的生长发育,而且影响作物品质和产量[5-8]。因此元素的形态、有效态分析越来越受到研究人员的重视[9-10]。通过土壤有效磷的测定,有助于了解土壤供应磷的情况,为合理使用磷肥及提高磷肥利用率提供依据[11-13]。通常所谓的土壤有效磷是指某一特定形态的磷,只是一个相对指标,但是其可用于说明土壤的供磷水平[14-15]。
目前,测定土壤有效磷的方法主要有Olsen法和Bray法[16]。Olsen法用碳酸氢钠溶液浸提土壤,Bray法用氟化铵-盐酸溶液浸提土壤。方法原理是浸提土壤后,分取浸提液加入显色剂,使用分光光度法测定浸提液中的磷含量。其中Bray法对酸性土壤效果良好,Olsen法对中性及石灰性土壤效果良好[17]。现行标准方法《土壤检测第7部分土壤有效磷的测定》(NY/T 1121.7—2014)和《森林土壤磷的测定》(LY/T 1232—2015)中Olsen法和Bray法的应用存在参数差异[18],主要差异在于酸性土壤的浸提时间、振荡温度等。目前常用的土壤有效态标准样品中,ASA1-6(GBW07412)、NSA1-6是依据GB/T 7853—1987定值,ASA1a至ASA6a、ASA-7至ASA-10是依据NY/T 1121.7—2006定值(GB/T 7853—1987、NY/T 1121.7—2006分别已被LY/T 1232—2015、NY/T 1121.7—2014替代)。已有文献报道了使用电感耦合等离子体发射光谱法(ICP-OES)测定浸提液中的有效磷[19-22],由于ICP-OES测定磷的灵敏度低,检出限高,达不到部分土壤样品的有效磷含量,因此分光光度法仍是测定土壤中有效磷的最优选择。该技术存在以下问题:①现有测定方法需浸提后过滤得到澄清待测液,过滤操作繁琐,且有些土壤沉淀颗粒细,极易穿透滤纸,速度慢效率低。②浸提液显色前必须先将pH调至指定值,再加钼锑抗显色剂才能发生特定显色反应。且该方法常使用二硝基酚(变色范围为无色pH 2.4~4.0黄色)作指示剂,由黄色调至恰变浅黄,而土壤浸提液多略带黄色的底色,尤其是有机质含量高的土壤,浸提滤液易呈现黄或褐色[23-25],导致调pH时颜色无法判断;且二硝基酚属于易制爆试剂,毒性强[26]。
针对以上问题,本文采用离心分离代替过滤;比较了不同浸提时间、不同浸提液酸度、不同酸碱指示剂等实验条件对检测结果的影响;使用溴酚蓝作指示剂调节pH,对比了溴酚蓝指示剂与标准方法的二硝基酚指示剂的检测结果。拟通过对现有方法进行优化创新,提高检测效率。
1. 实验部分
1.1 仪器及工作条件
分光光度计(北京普析通用仪器有限责任公司,型号T9CS),离心机(天津广丰科技有限公司,型号TD6M),恒温振荡培养箱(苏州培英实验设备有限公司,型号HZQ-X100)。
1.2 材料和主要试剂
磷标准溶液1000mg/L(国家有色金属及电子材料分析测试中心)。
溴酚蓝指示剂:称取0.20g溴酚蓝溶于100mL乙醇中。
盐酸(北京化学试剂研究所,BⅧ级);盐酸(国药集团化学试剂有限公司,光谱纯);氟化铵(Strem Chemicals,ACS);氟化铵(上海麦克林生化科技有限公司,ACS)。
酸性土壤:土壤有效态成分分析标准样品GBW07416、GBW07412(中国地质科学院地球物理地球化学勘查研究所);未知样A(野外采集,测定pH < 6.5)。
中性及石灰性土壤:土壤有效态成分分析标准样品GBW07413、GBW07414、GBW07414a(中国地质科学院地球物理地球化学勘查研究所);未知样B(野外采集,测定pH>6.5)。
其他试剂参考标准方法《土壤检测第7部分土壤有效磷的测定》(NY/T 1121.7—2014)配制,均采用市售分析纯。
1.3 实验条件
参考标准方法《土壤检测第7部分土壤有效磷的测定》(NY/T 1121.7—2014)。
酸性土壤:①待测液的制备:称取酸性土壤5.00g于200mL塑料浸提瓶中,加氟化铵(0.03mol/L)-盐酸(0.025mol/L)浸提液50.0mL,25℃以160r/min恒温振荡60min后,5000r/min离心5min。②移取上清液10mL于50mL比色管中,加入5.00mL硼酸(30g/L),加水,加入溴酚蓝指示剂2滴,用2mol/L盐酸和2mol/L氨水调节至试液刚呈黄色。③测定:加5.00mL钼锑抗显色剂,定容至50mL,摇匀,25℃以上放置30min后用分光光度计在700nm或880nm处进行吸光度测定,绘制标准曲线,计算出待测样品中有效磷含量。
中性或石灰性土壤:①待测液的制备:称取中性或石灰性土壤2.50g于200mL塑料浸提瓶中,加50.0mL碳酸氢钠浸提液(42g/L),25℃以160r/min恒温振荡30min后,5000r/min离心5min。②移取上清液10mL于50mL比色管中,加入溴酚蓝指示剂2滴,用0.5mol/L硫酸调节至试液刚呈黄色。③测定:加5.00mL钼锑抗显色剂,定容至50mL,摇匀,25℃以上放置30min后用分光光度计在700nm或880nm处进行吸光度测定,绘制标准曲线,计算出待测样品中有效磷含量。
2. 结果与讨论
2.1 酸性土壤浸提时间及土水比实验结果
用土壤有效态国家标准物质GBW07416开展条件实验,改变酸性土壤浸提时间及水土比,其他条件参考标准方法《土壤检测第7部分土壤有效磷的测定》(NY/T 1121.7—2014)。结果如图 1所示,氟化铵-盐酸偏提取酸性土壤时,随着浸提液比例降低,测定结果降低;随着提取时间增加,提取出的有效磷逐渐减少,与文献报道结果一致。王荣辉等[18]提出,可能是由于土壤体系自身缓冲作用,浸提时间延长,土壤体系pH升高,导致F-对Fe3+和Al3+的配合能力减弱,Fe-P和Al-P的释放量减少而引起。用GBW07416标准物质研究氟化铵-盐酸浸提时间与pH关系,结果如图 2所示,与文献[18]结论一致,且与不同浸提时间的测定结果一致。
本实验中使用标准方法的土水比1:10时,需浸提60min,才得到标样值结果。而中性及石灰性土壤可依据标准方法的实验条件测得相应的标样值。
2.2 溴酚蓝指示剂的对比实验
配制标准曲线0、1、5、10、20、30μg/50mL点,并选用国家标准物质GBW07412、GBW07414a、未知土壤样品A、未知土壤样品B及流程空白溶液,调节浸提液pH时分别使用二硝基酚和溴酚蓝作指示剂,用1cm比色池,比较两种方法的吸光度和检测结果。通过比较酸性土壤和中性、石灰性土壤标准曲线点的吸光度,结果表明用这两种指示剂调节pH后,加入显色剂,显色吸光度没有明显差异。表 1是分别用酸性土壤方法、中性和石灰性土壤方法分析对应的土壤未知样、标准物质、空白对比实验结果,可见使用溴酚蓝替代二硝基酚,检测结果完全一致,标准物质结果符合要求,说明本文优化方法可以适用于酸性、中性和石灰性土壤。使用溴酚蓝(变色范围黄色pH 3.0~4.6蓝色)作指示剂调至适宜pH值时颜色为黄色,磷酸根与钼锑抗显色剂形成蓝色配合物,两种颜色波长差异非常大,不干扰后续显色的测定。而且钼锑抗显色剂呈强酸性,加入后不会使待测液pH值向碱性变化而导致溴酚蓝显色剂变色影响实验结果。
表 1 使用不同pH指示剂测定土壤中有效磷结果对比Table 1. Comparison of analytical results of available phosphorus in soil extracted by different pH indicator不同酸碱性土壤 待测液调节pH指示剂 实验样品 吸光度 溶液中有效磷浓度
(μg/50mL)样品中有效磷测定值
(μg/g)有效磷标准值
(μg/g)酸性土壤 二硝基酚 GBW07412 0.245 22.305 22.30 21.2±3.6 未知样A 0.099 8.568 8.57 / 空白溶液 0.009 0.153 / / 溴酚蓝 GBW07412 0.266 23.648 23.65 21.2±3.6 未知样A 0.104 8.458 8.46 / BK 0.006 -0.070 / / 中性或石灰性土壤 二硝基酚 GBW07414a 0.140 14.721 29.44 29±3 未知样B 0.049 6.286 12.57 / 空白溶液 -0.017 0.263 / / 溴酚蓝 GBW07414a 0.116 14.248 28.50 29±3 未知样B 0.024 5.790 11.58 / 空白溶液 -0.020 0.013 / / 由于土壤样品的复杂性,偏提取得到的待测溶液可能含有未知物质,由于偏提取过程中化学反应的复杂性,土壤中的未知物有可能会干扰所用指示剂溴酚蓝的使用效果,但现有方法所用指示剂二硝基酚也存在这种未知性,本方法应用中尚未见不适用情况。
2.3 浸提液pH的影响
标准方法《土壤检测第7部分土壤有效磷的测定》(NY/T 1121.7—2014)中碳酸氢钠浸提液配制后,需用氢氧化钠溶液调节pH至8.5,并定期监测浸提液pH值。选用两个标准物质,配制后直接使用并与调节pH后的浸提液分别测定,有效磷结果见表 2,碳酸氢钠浸提液pH值对结果影响很大,配制后溶液pH为8.22,其浸提检测结果明显偏低。
表 2 碳酸氢钠浸提液pH对有效磷检测结果的影响Table 2. Effect of NaHCO3 extraction with different pH on analytical results of available phosphorus标准物质编号 有效磷测定值(μg/g) 有效磷标准值
(μg/g)未调节pH 调节pH至8.5 GBW07413 14.20 17.49 18.3±2 GBW07414 10.26 12.71 13.8±2.3 氟化铵-盐酸浸提液的pH在标准方法中未作强调,部分文献[27]中偶见pH 1.5~2.0。如2.1章节所述,由于本研究中的浸提时间与行业标准方法《土壤检测第7部分土壤有效磷的测定》(NY/T 1121.7—2014)不一致,且碳酸氢钠碱性浸提液pH对实验结果有主要影响,为了了解酸性浸提液pH的影响程度,实验中尝试用不同厂家的氟化铵及盐酸试剂,按标准方法配制的酸性浸提液pH值为2.7~3.2,测定结果没有明显差异。实验表明将氟化铵-盐酸浸提液pH调节至2后,浸提出大量的磷,明显不适用于有效磷的测定,因此氟化铵-盐酸浸提液配制后可直接使用。实验中发现,氟化铵-盐酸浸提液放置一个月,pH值由2.72略升高至2.79,其浸提GBW07416一小时后溶液pH值为5.04,也比现配的溶液浸提后的pH值(4.90)高。虽检测结果未见明显差异,但浸提液的pH值可能会影响浸提能力,最好每周配制。
以上实验表明,碳酸氢钠浸提液配制后必须调节pH至8.5,氟化铵-盐酸浸提液配制后可直接使用。
2.4 已浸提溶液的存放时间
《森林土壤有效磷的测定》(LY/T 1233—1999)提到浸提后溶液的保存时间,需24h之内测定。本研究测定了两个不同含量水平的酸性土壤样品,结果见表 3,有效磷含量在2μg/g左右的样品,浸提液滤液放置3天检测结果下降约7.57%,放置5天检测结果下降约9.17%;有效磷含量在15μg/g左右的标准样品(GBW07416),浸提液滤液放置3天检测结果下降约0.43%,放置5天检测结果下降约13.20%。可见浸提液久置后有效磷会损失,其含量下降程度与样品及含量有关,最好在浸提当天完成检测。
表 3 已浸提溶液的存放时间对有效磷检测结果的影响Table 3. Effect of extracted solution with different storage time on analytical results of available phosphorus样品 放置不同时间有效磷测定值(μg/g) 有效磷测定值下降比例(%) 0d 3d 5d 3d 5d 未知样品 2.54 2.34 2.30 -7.57 -9.17 GBW07416 17.05 16.98 14.80 -0.43 -13.20 2.5 方法检出限和精密度
选用酸性土壤有效态标准物质GBW07416和碱性土壤有效态标准物质GBW07414,分别平行偏提取7份,检测结果见表 4。GBW07416检测结果平均值为17.2μg/g,GBW07414检测结果平均值为12.5μg/g,均在标准值范围内,准确度和相对标准偏差符合要求。依据《环境监测分析方法标准制修订技术导则》(HJ 168—2010)附录A.1.2,使用3cm比色皿时,与0.01吸光度对应的浓度值作为检出限,对应曲线浓度点约为0.5μg/50mL。称样量为5.0g分取10mL浸提液时,检出限为0.5μg/g;称样量为2.5g分取10mL浸提液时,检出限为1.0μg/g。
表 4 方法精密度Table 4. Precision tests of the method参数 GBW07416 GBW07414 有效磷测定平均值(μg/g) 17.2 12.5 有效磷标准值(μg/g) 14.8±3.1 13.8±2.3 标准偏差(n=7) 0.5907 0.2323 相对标准偏差(%,n=7) 3.44 1.86 3. 结论
本研究探讨了测定土壤中有效磷标准方法中的实验条件对检测结果的影响,以及酸性土壤浸提时间及土水比对测定结果的影响。结果表明,酸性土壤在1:10土水比条件下,需浸提60min可得到标样值结果,而碱性土壤可依据标准方法实验条件得到标样值结果;同时探讨了浸提液pH的影响,碳酸氢钠浸提液需调pH至8.5,否则结果明显偏低,氟化铵-盐酸浸提液不可调pH且不可久置;对于已浸提溶液的存放时间,最好在浸提当天完成检测。
本研究采用溴酚蓝指示剂替代标准方法的二硝基酚指示剂,标准方法使用的二硝基酚指示剂颜色突变不明显,易受浸提液底色影响,且易制爆、毒性强;而溴酚蓝指示剂调节pH时突变明显,不受浸提液底色干扰,优于标准方法的二硝基酚指示剂。基于两种指示剂的吸光度和检测结果完全一致,表明溴酚蓝指示剂可适用于土壤中有效磷的测定。
-
表 1 不同国家土壤Cr污染现状
Table 1 Current situation of Cr pollution in different countries
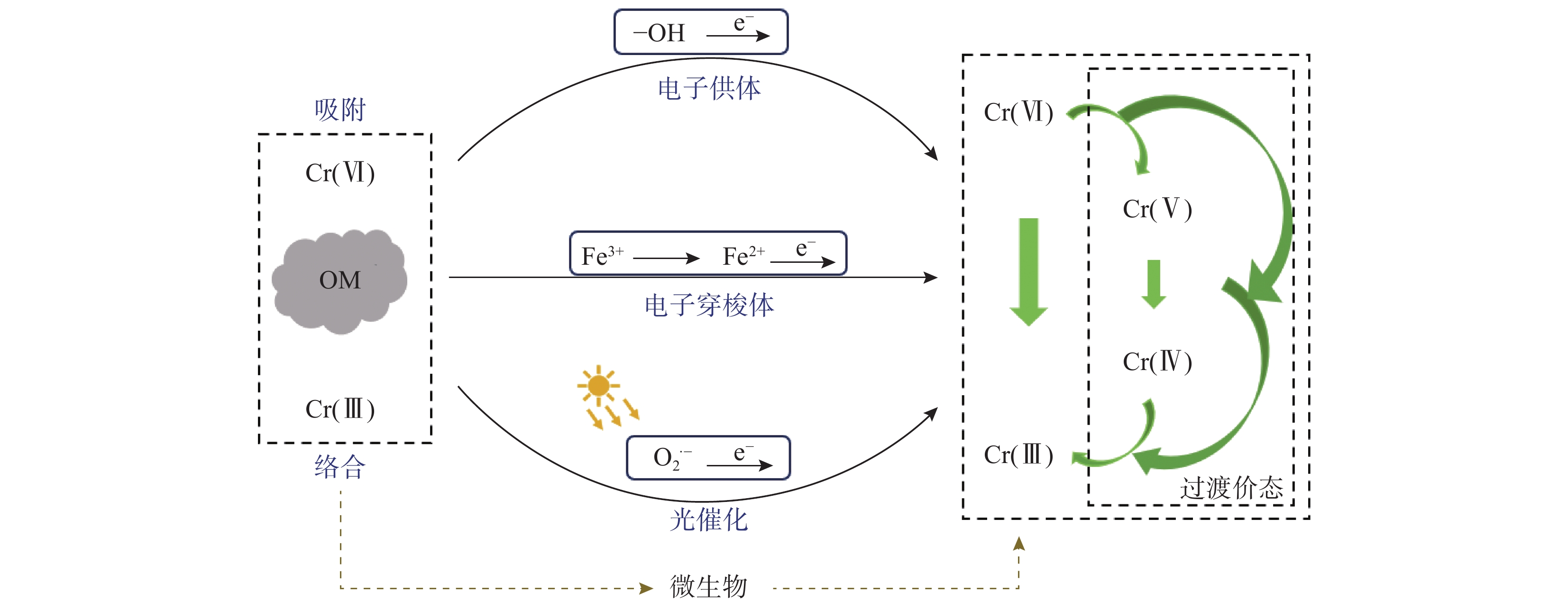
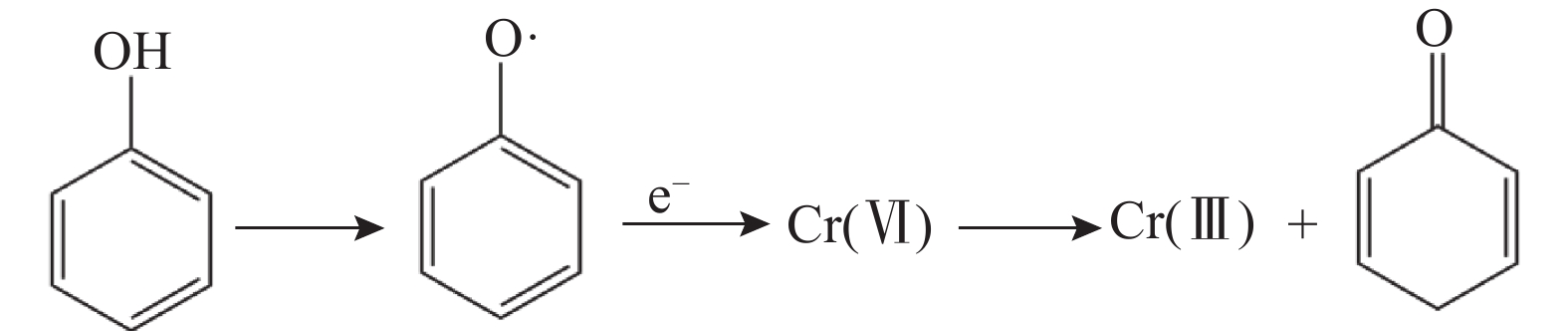
研究场地 Cr(Ⅲ)含量
(mg/kg)Cr(Ⅵ)含量
(mg/kg)总Cr含量
(mg/kg)对照标准 是否污染
地下水埃塞俄比亚某制革厂周边
污染区域土壤[19]791.5~1811.4 0.621~0.973 792.47~1812.21 制革厂90公里外某处未受污染土壤样品:Cr(Ⅲ)浓度0.18mg/kg,
Cr(Ⅵ)未检出,总Cr浓度0.18mg/kg是 意大利南部一处往年被长期非法掩埋皮革工业废物的农田[20] — 0.15~11.18 48~6831 意大利议会制定的关于土壤中Cr(Ⅵ)筛选值2mg/kg,
总Cr筛选值150mg/kg未评估 中国重庆某铬盐生产场地
遗留旧址[21]— 168.8~203.4 — 《土壤环境质量标准》
(GB 36600—2018)限值5.7mg/kg未评估 中国天津原同生化工厂残渣垃圾堆放场地及周边表层土壤[22] — — 8571.4~10711.4 《土壤环境质量标准》
(GB 15618—2018) 限值250mg/kg是 中国宁夏回族自治区某煤化
化工厂周边土壤[23]— 6.480~11.750 73.800~107.080[24] 中国土壤Cr(Ⅵ)背景值6.100mg/kg;宁夏土壤Cr(Ⅵ)背景值6.000mg/kg 未评估 注:“—”表示数据缺失。 -
[1] Sharma A, Kapoor D, Wang J, et al. Chromium bioaccumulation and its impacts on plants: An overview[J]. Plants-Basel, 2020, 9(1): 100. doi: 10.3390/plants9010100
[2] Pourret O, Hursthouse A. It’s time to replace the term “Heavy Metals”with “Potentially Toxic Elements” when reporting environmental research[J]. International Journal of Environmental Research and Public Health, 2019, 16(22): 4446. doi: 10.3390/ijerph16224446
[3] Rashid A, Schutte B J, Ulery A, et al. Heavy metal contamination in agricultural soil: Environmental pollutants affecting crop health[J]. Agronomy-Basel, 2023, 13(6): 1521. doi: 10.3390/agronomy13061521
[4] Rinklebe J, Antoniadis V, Shaheen S M, et al. Health risk assessment of potentially toxic elements in soils along the Central Elbe River, Germany[J]. Environment International, 2019, 126: 76−88. doi: 10.1016/j.envint.2019.02.011
[5] 林晓梅, 曹玉莹, 赵上勇, 等. 激光诱导击穿光谱技术对土壤中重金属元素Cr的定量分析[J]. 光谱学与光谱分析, 2021, 41(3): 875−879 doi: 10.3964/j.issn.1000-0593(2021)-0875-05 Lin X M, Cao Y Y, Zhao S Y, et al. Quantitative analysis of Cr in soil by laser-induced breakdown spectroscopy[J]. Spectroscopy and Spectral Analysis, 2021, 41(3): 875−879. doi: 10.3964/j.issn.1000-0593(2021)-0875-05
[6] Mortada W I, El-Naggar A, Mosa A, et al. Biogeochemical behaviour and toxicology of chromium in the soil-water-human nexus: A review[J]. Chemosphere, 2023, 331: 138804. doi: 10.1016/j.chemosphere.2023.138804
[7] Prado C, Ponce S C, Pagano E, et al. Differential physiological responses of two Salvinia species to hexavalent chromium at a glance[J]. Aquatic Toxicology, 2016, 175: 213−221. doi: 10.1016/j.aquatox.2016.03.027
[8] Wei Y, Usman M, Farooq M, et al. Removing hexavalent chromium by nano zero-valent iron loaded on attapulgite[J]. Water Air and Soil Pollution, 2022, 233(2): 48. doi: 10.1007/s11270-022-05513-z
[9] Mongaa A, Fulkea A B, Dasguptab D. Recent developments in essentiality of trivalent chromium and toxicity of hexavalent chromium: Implications on human health and remediation strategies[J]. Journal of Hazardous Materials Advances, 2022, 7: 100113. doi: 10.1016/j.hazadv.2022.100113
[10] den Braver-Sewradj S P, van Benthem J, Staal Y C M, et al. Occupational exposure to hexavalent chromium. Part Ⅱ. Hazard assessment of carcinogenic effects[J]. Regulatory Toxicology and Pharmacology, 2021, 126: 105045. doi: 10.1016/j.yrtph.2021.105045
[11] Yan X, Yan Z H, Zhu X Z, et al. Comparing different strategies for Cr(Ⅵ) bioremediation: Bioaugmentation, biostimulation, and bioenhancement[J]. Sustainability, 2023, 15(16): 12522. doi: 10.3390/su151612522
[12] Sayed D, Alturki A A, Farag H, et al. A novel rotating fixed bed batch reactor for hexavalent chromium reduction[J]. Journal of Ecological Engineering, 2022, 23(11): 273−280. doi: 10.12911/22998993/154061
[13] Kang Z, Gao H, Ma X, et al. Fe-Ni/MWCNTs nano-composites for hexavalent chromium reduction in aqueous environment[J]. Molecules, 2023, 28(11): 4412. doi: 10.3390/molecules28114412
[14] Guo H, Chen Y, Hu H, et al. High hexavalent chromium concentration in groundwater from a deep aquifer in the Baiyangdian Basin of the North China Plain[J]. Environmental Science & Technology, 2020, 54(16): 10068−10077. doi: 10.1021/acs.est.0c02357
[15] Deng L Y, Liu F, Ding Z C, et al. Effect of natural organic matter on Cr(Ⅵ) reduction by reduced nontronite[J]. Chemical Geology, 2023, 615: 121198. doi: 10.1016/j.chemgeo.2022.121198
[16] Enbaia S, Eswayah A, Hondow N, et al. Detoxification, active uptake, and intracellular accumulation of chromium species by a methane-oxidizing bacterium[J]. Applied and Environmental Microbiology, 2021, 87(2): e00947−e00920. doi: 10.1128/AEM.00947-20
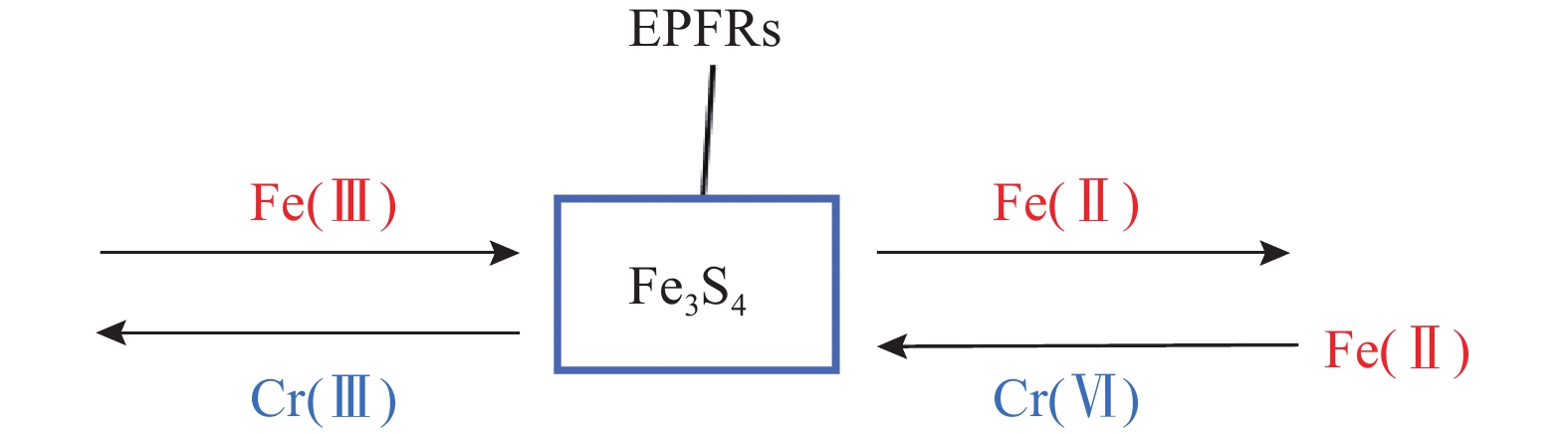
[17] Pei Y, Yang Y, Chen L, et al. Remediation of chromium-contaminated soil in semi-arid areas by combined chemical reduction and stabilization[J]. Environmental Pollutants and Bioavailability, 2023, 35(1): 2157332. doi: 10.1080/26395940.2022.2157332
[18] Li S, Xie Y, Jiang S, et al. Biochar decreases Cr toxicity and accumulation in sunflower grown in Cr(Ⅵ)-polluted soil[J]. Toxics, 2023, 11(9): 787. doi: 10.3390/toxics11090787
[19] Gezahegn A M, Feyessa F F, Tekeste E A, et al. Chromium laden soil, water, and vegetables nearby tanning industries: Speciation and spatial distribution[J]. Journal of Chemistry, 2021, 2021: 5531349. doi: 10.1155/2021/5531349
[20] Caporale A G, Agrelli D, Rodríguez-González P, et al. Hexavalent chromium quantification by isotope dilution mass spectrometry in potentially contaminated soils from South Italy[J]. Chemosphere, 2019, 233: 92−100. doi: 10.1016/j.chemosphere.2019.05.212
[21] Zhong W, Bai W, Li G. Reduction of hexavalent chromium from soil of the relocated factory area with rice straw hydrothermal carbon modified by nano zero-valent iron (nZVI)[J]. International Journal of Environmental Research and Public Health, 2023, 20(4): 3089. doi: 10.3390/ijerph20043089
[22] Liu Y, Li Y, Hu Y C, et al. Adsorption characteristics and transport behavior of Cr(Ⅵ) in shallow aquifers surrounding a chromium ore processing residue (copr) dumpsite[J]. Journal of Chemistry, 2019, 2019: 4932837. doi: 10.1155/2019/4932837
[23] Zhang K, Yang J, Wang Y, et al. All-region human health risk assessment of Cr(Ⅵ) in a coal chemical plant based on Kriging[J]. Polish Journal of Environmental Studies, 2020, 29(1): 429−439. doi: 10.15244/pjoes/99226
[24] Zhang K, Qiang C D, Liu J. Spatial distribution characteristics of heavy metals in the soil of coal chemical industrial areas[J]. Journal of Soils and Sediments, 2018, 18(5): 2044−2052. doi: 10.1007/s11368-018-1972-9
[25] Li Y, Pan S, Wang L, et al. Soil chromium accumulation in industrial regions across China: Pollution and health risk assessment, spatial pattern, and temporal trend (2002—2021)[J]. Toxics, 2023, 11(4): 363. doi: 10.3390/toxics11040363
[26] Xiang J, Xu P, Chen W, et al. Pollution characteristics and health risk assessment of heavy metals in agricultural soils over the past five years in Zhejiang, Southeast China[J]. International Journal of Environmental Research and Public Health, 2022, 19(22): 14642. doi: 10.3390/ijerph192214642
[27] Wu D, Liu H, Wu J, et al. Bi-directional pollution characteristics and ecological health risk assessment of heavy metals in soil and crops in Wanjiang Economic Zone, Anhui Province, China[J]. International Journal of Environmental Research and Public Health, 2022, 19(15): 9669. doi: 10.3390/ijerph19159669
[28] Wu Z, Zhang D, Xia T, et al. Characteristics, sources and risk assessments of heavy metal pollution in soils of typical chlor-alkali residue storage sites in Northeastern China[J]. PLOS One, 2022, 17(9): e0273434. doi: 10.1371/journal.pone.0273434
[29] Li X, Zhang J, Ma J, et al. Status of chromium accumulation in agricultural soils across China (1989—2016)[J]. Chemosphere, 2020, 256: 127036. doi: 10.1016/j.chemosphere.2020.127036
[30] Kanagaraj G, Elango L. Chromium and fluoride contamination in groundwater around leather tanning industries in Southern India: Implications from stable isotopic ratio δ53Cr/δ52Cr, geochemical and geostatistical modelling[J]. Chemosphere, 2019, 220: 943−953. doi: 10.1016/j.chemosphere.2018.12.105
[31] Chrysochoou M, Theologou E, Bompoti N, et al. Occurrence, origin and transformation processes of geogenic chromium in soils and sediments[J]. Current Pollution Reports, 2016, 2(4): 224−235. doi: 10.1007/s40726-016-0044-2
[32] Kierczak J, Pietranik A, Pedziwiatr A. Ultramafic geoecosystems as a natural source of Ni, Cr, and Co to the environment: A review[J]. Science of the Total Environment, 2021, 755: 142620. doi: 10.1016/j.scitotenv.2020.142620
[33] Briffa J, Sinagra E, Blundell R. Heavy metal pollution in the environment and their toxicological effects on humans[J]. Heliyon, 2020, 6(9): e04691. doi: 10.1016/j.heliyon.2020.e04691
[34] Coetzee J J, Bansal N, Chirwa E M N. Chromium in environment, its toxic effect from chromite-mining and ferrochrome industries, and its possible bioremediation[J]. Exposure and Health, 2020, 12(1): 51−62. doi: 10.1007/s12403-018-0284-z
[35] Pushkar B, Sevak P, Parab S, et al. Chromium pollution and its bioremediation mechanisms in bacteria: A review[J]. Journal of Environmental Management, 2021, 287: 112279. doi: 10.1016/j.jenvman.2021.112279
[36] 贾琼琳, 韩俊艳, 何丹. 土壤中重金属铬污染及其治理方法的研究进展[C]//中国环境科学学会2021年科学技术年会——环境工程技术创新与应用分会场. 北京: 中国环境科学学会, 2021: 354−357, 380. Jia Q L, Han J Y, He D. Research progress of heavy metal chromium pollution in soil and its control methods[C]//China Environmental Science Society 2021 Science and Technology Annual Conference—Environmental Engineering Technology Innovation and Application Sub-forum. Beijing: Chinese Society for Environmental Sciences, 2021: 354−357, 380.
[37] Gupta D K, Chatterjee S, Datta S, et al. Role of phosphate fertilizers in heavy metal uptake and detoxification of toxic metals[J]. Chemosphere, 2014, 108: 134−144. doi: 10.1016/j.chemosphere.2014.01.030
[38] 耿源濛, 张传兵, 张勇, 等. 我国城市污泥中重金属的赋存形态与生态风险评价[J]. 环境科学, 2021, 42(10): 4834−4843. doi: 10.13227/j.hjkx.202101145 Geng Y M, Zhang C B, Zhang Y, et al. Speciation and ecological risk assessment of heavy metal(loid)s in the municipal sewage sludge of China[J]. Environmental Science, 2021, 42(10): 4834−4843. doi: 10.13227/j.hjkx.202101145
[39] 白宇明, 李永利, 周文辉, 等. 典型工业城市土壤重金属元素形态特征及生态风险评估[J]. 岩矿测试, 2022, 41(4): 632−641. doi: 10.15898/j.cnki.11-2131/td.202109030113 Bai Y M, Li Y L, Zhou W H, et al. Speciation characteristics and ecological risk assessment of heavy metal elements in soils of typical industrial city[J]. Rock and Mineral Analysis, 2022, 41(4): 632−641. doi: 10.15898/j.cnki.11-2131/td.202109030113
[40] Ali W, Mao K, Zhang H, et al. Comprehensive review of the basic chemical behaviours, sources, processes, and endpoints of trace element contamination in paddy soil-rice systems in rice-growing countries[J]. Journal of Hazardous Materials, 2020, 397: 122720. doi: 10.1016/j.jhazmat.2020.122720
[41] Bai J, Xun P, Morris S, et al. Chromium exposure and incidence of metabolic syndrome among American young adults over a 23-year follow-up: The CARDIA trace element study[J]. Scientific Reports, 2015, 5: 15606. doi: 10.1038/srep15606
[42] Sharma P, Singh S P, Parakh S K, et al. Health hazards of hexavalent chromium (Cr(Ⅵ)) and its microbial reduction[J]. Bioengineered, 2022, 13(3): 4923−4938. doi: 10.1080/21655979.2022.2037273
[43] Ulhassan Z, Gill R A, Huang H, et al. Selenium mitigates the chromium toxicityin Brassicca napus L. by ameliorating nutrients uptake, amino acids metabolism and antioxidant defense system[J]. Plant Physiology and Biochemistry, 2019, 145: 142−152. doi: 10.1016/j.plaphy.2019.10.035
[44] Kundu D, Dey S, Raychaudhuri S S. Chromium(Ⅵ) induced stress response in the plant Plantago ovata Forsk in vitro[J]. Genes and Environment, 2018, 40: 21. doi: 10.1186/s41021-018-0109-0
[45] Aziz S, Altaf J, Khalil A, et al. Human cancer risk due to chromium and its bioaccumulation in physids in Central Punjab, Pakistan[J]. Environmental Science and Pollution Research, 2023, 30(29): 74223−74235. doi: 10.1007/s11356-023-27664-0
[46] Banu S K, Stanley J A, Taylor R J, et al. Sexually dimorphic impact of chromium accumulation on human placental oxidative stress and apoptosis[J]. Toxicological Sciences, 2018, 161(2): 375−387. doi: 10.1093/toxsci/kfx224
[47] Guo S, Xiao C, Zhou N, et al. Speciation, toxicity, microbial remediation and phytoremediation of soil chromium contamination[J]. Environmental Chemistry Letters, 2020, 19(2): 1413−1431. doi: 10.1007/s10311-020-01114-6
[48] Ertani A, Mietto A, Borin M, et al. Chromium in agricultural soils and crops: A review[J]. Water Air and Soil Pollution, 2017, 228(5): 190. doi: 10.1007/s11270-017-3356-y
[49] Wani K I, Naeem M, Aftab T. Chromium in plant-soil nexus: Speciation, uptake, transport and sustainable remediation techniques[J]. Environmental Pollution, 2022, 315: 120350. doi: 10.1016/j.envpol.2022.120350
[50] Xiao L, Guan D, Chen Y, et al. Distribution and availability of heavy metals in soils near electroplating factories[J]. Environmental Science and Pollution Research, 2019, 26(22): 22596−22610. doi: 10.1007/s11356-019-04706-0
[51] Xiao W, Ye X, Yang X, et al. Effects of alternating wetting and drying versus continuous flooding on chromium fate in paddy soils[J]. Ecotoxicology and Environmental Safety, 2015, 113: 439−445. doi: 10.1016/j.ecoenv.2014.12.030
[52] Shahid M, Shamshad S, Rafiq M, et al. Chromium speciation, bioavailability, uptake, toxicity and detoxification in soil-plant system: A review[J]. Chemosphere, 2017, 178: 513−533. doi: 10.1016/j.chemosphere.2017.03.074
[53] Liang J, Huang X, Yan J, et al. A review of the formation of Cr(Ⅵ) via Cr(Ⅲ) oxidation in soils and groundwater[J]. Science of the Total Environment, 2021, 774: 145762. doi: 10.1016/j.scitotenv.2021.145762
[54] Li B, Yang J X, Sun W T, et al. Carbonization of plant residues decreased their capability of reducing hexavalent chromium in soils[J]. Water Air and Soil Pollution, 2019, 230(12): 300. doi: 10.1007/s11270-019-4353-0
[55] Zhang Z, Ren J, Liang J, et al. New insight into the natural detoxification of Cr(Ⅵ) in Fe-rich surface soil: Crucial role of photogenerated silicate-bound Fe(Ⅱ)[J]. Environmental Science & Technology, 2023, 57(50): 21370−21381. doi: 10.1021/acs.est.3c05767
[56] Hao Y, Ma H, Wang Q, et al. Complexation behaviour and removal of organic-Cr(Ⅲ) complexes from the environment: A review[J]. Ecotoxicology and Environmental Safety, 2022, 240: 113676. doi: 10.1016/j.ecoenv.2022.113676
[57] Bokare A D, Choi W. Advanced oxidation process based on the Cr(Ⅲ)/Cr(Ⅵ) redox cycle[J]. Environmental Science & Technology, 2011, 45(21): 9332−9338. doi: 10.1021/es2021704
[58] Jiang B, Gong Y, Gao J, et al. The reduction of Cr(Ⅵ) to Cr(Ⅲ) mediated by environmentally relevant carboxylic acids: State-of-the-art and perspectives[J]. Journal of Hazardous Materials, 2019, 365: 205−226. doi: 10.1016/j.jhazmat.2018.10.070
[59] Zheng C, Yang Z, Si M, et al. Application of biochars in the remediation of chromium contamination: Fabrication, mechanisms, and interfering species[J]. Journal of Hazardous Materials, 2021, 407: 124376. doi: 10.1016/j.jhazmat.2020.124376
[60] Shi Y, Shan R, Lu L, et al. High-efficiency removal of Cr(Ⅵ) by modified biochar derived from glue residue[J]. Journal of Cleaner Production, 2020, 254: 119935. doi: 10.1016/j.jclepro.2019.119935
[61] Li H, Wang J, Zhao B, et al. The role of major functional groups: Multi-evidence from the binding experiments of heavy metals on natural fulvic acids extracted from lake sediments[J]. Ecotoxicology and Environmental Safety, 2018, 162: 514−520. doi: 10.1016/j.ecoenv.2018.07.038
[62] Xu J, Dai Y, Shi Y, et al. Mechanism of Cr(Ⅵ) reduction by humin: Role of environmentally persistent free radicals and reactive oxygen species[J]. Science of the Total Environment, 2020, 725: 138413. doi: 10.1016/j.scitotenv.2020.138413
[63] Zhang J, Yin H, Wang H, et al. Reduction mechanism of hexavalent chromium by functional groups of undissolved humic acid and humin fractions of typical black soil from Northeast China[J]. Environmental Science and Pollution Research, 2018, 25(17): 16913−16921. doi: 10.1007/s11356-018-1878-5
[64] Xu Z, Xu X, Zhang Y, et al. Pyrolysis-temperature depended electron donating and mediating mechanisms of biochar for Cr(Ⅵ) reduction[J]. Journal of Hazardous Materials, 2020, 388: 121794. doi: 10.1016/j.jhazmat.2019.121794
[65] Wang X, Xu J, Liu J, et al. Mechanism of Cr(Ⅵ) removal by magnetic greigite/biochar composites[J]. Science of the Total Environment, 2020, 700: 134414. doi: 10.1016/j.scitotenv.2019.134414
[66] Odinga E S, Waigi M G, Gudda F O, et al. Occurrence, formation, environmental fate and risks of environmentally persistent free radicals in biochars[J]. Environment International, 2020, 134: 105172. doi: 10.1016/j.envint.2019.105172
[67] Fan Z, Zhang Q, Gao B, et al. Removal of hexavalent chromium by biochar supported nZVI composite: Batch and fixed-bed column evaluations, mechanisms, and secondary contamination prevention[J]. Chemosphere, 2019, 217: 85−94. doi: 10.1016/j.chemosphere.2018.11.009
[68] 陈壮, 梁媛, 赵奔, 等. 改性生物炭对Cr(Ⅵ)的吸附特性研究[J]. 复旦学报(自然科学版), 2021, 60(6): 779−788. doi: 10.15943/j.cnki.fdxb-jns.2021.06.007 Chen Z, Liang Y, Zhao B, et al. Adsorption characteristics and mechanism of modified biochar to Cr(Ⅵ)[J]. Journal of Fudan University (Natural Science), 2021, 60(6): 779−788. doi: 10.15943/j.cnki.fdxb-jns.2021.06.007
[69] Li K, Huang Z, Zhu S, et al. Removal of Cr(Ⅵ) from water by a biochar-coupled g-C3N4 nanosheets composite and performance of a recycled photocatalyst in single and combined pollution systems[J]. Applied Catalysis B-Environmental, 2019, 243: 386−396. doi: 10.1016/j.apcatb.2018.10.052
[70] Peng X X, Gai S, Cheng K, et al. Roles of humic substances redox activity on environmental remediation[J]. Journal of Hazardous Materials, 2022, 435: 129070. doi: 10.1016/j.jhazmat.2022.129070
[71] Zhu S, Huang X, Yang X, et al. Enhanced transformation of Cr(Ⅵ) by heterocyclic-N within nitrogen-doped biochar: Impact of surface modulatory persistent free radicals (PFRs)[J]. Environmental Science & Technology, 2020, 54(13): 8123−8132. doi: 10.1021/acs.est.0c02713
[72] Fang G, Liu C, Wang Y, et al. Photogeneration of reactive oxygen species from biochar suspension for diethyl phthalate degradation[J]. Applied Catalysis B-Environmental, 2017, 214: 34−45. doi: 10.1016/j.apcatb.2017.05.036
[73] Yu Y, An Q, Jin L, et al. Unraveling sorption of Cr(Ⅵ) from aqueous solution by FeCl3 and ZnCl2-modified corn stalks biochar: Implicit mechanism and application[J]. Bioresource Technology, 2020, 297: 122466. doi: 10.1016/j.biortech.2019.122466
[74] Geng A, Xu L, Gan L, et al. Using wood flour waste to produce biochar as the support to enhance the visible-light photocatalytic performance of BiOBr for organic and inorganic contaminants removal[J]. Chemosphere, 2020, 250: 126291. doi: 10.1016/j.chemosphere.2020.126291
[75] Wang T, Liu S, Mao W, et al. Novel Bi2WO6 loaded N-biochar composites with enhanced photocatalytic degradation of rhodamine B and Cr(Ⅵ)[J]. Journal of Hazardous Materials, 2020, 389: 121827. doi: 10.1016/j.jhazmat.2019.121827
[76] Alsaiari M. Biomass-derived active carbon (AC) modified TiO2 photocatalyst for efficient photocatalytic reduction of chromium(Ⅵ) under visible light[J]. Arabian Journal of Chemistry, 2021, 14(8): 103258. doi: 10.1016/j.arabjc.2021.103258
[77] Jalili B, Sadegh-Zadeh F, Jabari-Giashi M, et al. Lead bioimmobilization in contaminated mine soil by Aspergillus niger SANRU[J]. Journal of Hazardous Materials, 2020, 393: 122375. doi: 10.1016/j.jhazmat.2020.122375
[78] Hussain S, Maqbool Z, Shahid M, et al. Simultaneous removal of reactive dyes and hexavalent chromium by a metal tolerant pseudomonas sp. Ws-d/183 harboring plant growth promoting traits[J]. International Journal of Agriculture and Biology, 2020, 23(2): 241−252. doi: 10.17957/IJAB/15.1282
[79] Tariq M, Waseem M, Rasool M H, et al. Isolation and molecular characterization of the indigenous Staphylococcus aureus strain K1 with the ability to reduce hexavalent chromium for its application in bioremediation of metal-contaminated sites[J]. Peerj, 2019, 7: e7726. doi: 10.7717/peerj.7726
[80] Pattnaik S, Dash D, Mohapatra S, et al. Improvement of rice plant productivity by native Cr(VI) reducing and plant growth promoting soil bacteria Enterobacter cloacae[J]. Chemosphere, 2020, 240: 124895. doi: 10.1016/j.chemosphere.2019.124895
[81] Sha C Y, Wu J, Wu J Q, et al. Effects of different fertilizers on soil microbial diversity during long-term fertilization of a corn field in Shanghai, China[J]. Diversity-Basel, 2023, 15(1): 78. doi: 10.3390/d15010078
[82] Jin Q, Zhang Y, Wang Q, et al. Effects of potassium fulvic acid and potassium humate on microbial biodiversity in bulk soil and rhizosphere soil of Panax ginseng[J]. Microbiological Research, 2022, 254: 126914. doi: 10.1016/j.micres.2021.126914
[83] Tang X, Huang Y, Li Y, et al. Study on detoxification and removal mechanisms of hexavalent chromium by microorganisms[J]. Ecotoxicology and Environmental Safety, 2021, 208: 111699. doi: 10.1016/j.ecoenv.2020.111699
[84] Gu B H, Chen J. Enhanced microbial reduction of Cr(Ⅵ) and U(Ⅵ) by different natural organic matter fractions[J]. Geochimica et Cosmochimica Acta, 2003, 67(19): 3575−3582. doi: 10.1016/S0016-7037(3)00162-5
[85] Chen Y, Wu H, Sun P, et al. Remediation of chromium-contaminated soil based on bacillus cereus WHX-1 immobilized on biochar: Cr(Ⅵ) transformation and functional microbial enrichment[J]. Frontiers in Microbiology, 2021, 12: 641913. doi: 10.3389/fmicb.2021.641913
[86] Huang X N, Min D, Liu D F, et al. Formation mechanism of organo-chromium(Ⅲ) complexes from bioreduction of chromium(Ⅵ) by Aeromonas hydrophila[J]. Environment International, 2019, 129: 86−94. doi: 10.1016/j.envint.2019.05.016
[87] Tan H, Wang C, Zeng G, et al. Bioreduction and biosorption of Cr(Ⅵ) by a novel Bacillus sp. CRB-B1 strain[J]. Journal of Hazardous Materials, 2020, 386: 121628. doi: 10.1016/j.jhazmat.2019.121628
[88] Zhuang L, Li Q, Chen J, et al. Carbothermal preparation of porous carbon-encapsulated iron composite for the removal of trace hexavalent chromium[J]. Chemical Engineering Journal, 2014, 253: 24−33. doi: 10.1016/j.cej.2014.05.038
[89] Shaheen S M, Niazi N K, Hassan N E E, et al. Wood-based biochar for the removal of potentially toxic elements in water and wastewater: A critical review[J]. International Materials Reviews, 2019, 64(4): 216−247. doi: 10.1080/09506608.2018.1473096
[90] Liu W, Jin L, Xu J, et al. Insight into pH dependent Cr(Ⅵ) removal with magnetic Fe3S4[J]. Chemical Engineering Journal, 2019, 359: 564−571. doi: 10.1016/j.cej.2018.11.192
[91] Lee S, Roh Y, Koh D C. Oxidation and reduction of redox-sensitive elements in the presence of humic substances in subsurface environments: A review[J]. Chemosphere, 2019, 220: 86−97. doi: 10.1016/j.chemosphere.2018.11.143
[92] Zhu S, Huang X, Wang D, et al. Enhanced hexavalent chromium removal performance and stabilization by magnetic iron nanoparticles assisted biochar in aqueous solution: Mechanisms and application potential[J]. Chemosphere, 2018, 207: 50−59. doi: 10.1016/j.chemosphere.2018.05.046
[93] Gustafsson J P, Persson I, Oromieh A G, et al. Chromium(Ⅲ) complexation to natural organic matter: Mechanisms and modeling[J]. Environmental Science & Technology, 2014, 48(3): 1753−1761. doi: 10.1021/es404557e
[94] 刘爱科, 顾梦琪, 魏书斋, 等. 蒽醌-2,6-二磺酸(AQDS)强化厌氧降解直接蓝15[J]. 净水技术, 2019, 38(2): 63−68. doi: 10.15890/j.cnki.jsjs.2019.02.011 Liu A K, Gu M Q, Wei S Z, et al. Enhanced anaerobic degradation of direct blue 15 by anthraquinone-2,6-disulfonate (AQDS)[J]. Water Purification Technology, 2019, 38(2): 63−68. doi: 10.15890/j.cnki.jsjs.2019.02.011
[95] Tomaszewski E J, Ginder-Vogel M. Decreased electron transfer between Cr(Ⅵ) and AH2DS in the presence of goethite[J]. Journal of Environmental Quality, 2018, 47(1): 139−146. doi: 10.2134/jeq2017.08.0316
[96] Langer M, Jamal M U, Conklin A, et al. Chromium removal in the presence of NOM during Fe(Ⅱ) reductive precipitation for drinking water treatment[J]. Water, 2022, 14(18): 2903. doi: 10.3390/w14182903
[97] Wittbrodt P R, Palmer C D. Effect of temperature, ionic strength, background electrolytes, and Fe(Ⅲ) on the reduction of hexavalent chromium by soil humic substances[J]. Environmental Science & Technology, 1996, 30(8): 2470−2477. doi: 10.1021/es950731c
[98] Song C X, Sun S Q, Wang J T, et al. Applying fulvic acid for sediment metals remediation: Mechanism, factors, and prospect[J]. Frontiers in Microbiology, 2023, 13: 1084097. doi: 10.3389/fmicb.2022.1084097
[99] Bao Z J, Feng H Y, Tu W Y, et al. Method and mechanism of chromium removal from soil: A systematic review[J]. Environmental Science and Pollution Research, 2022, 29(24): 35501−35517. doi: 10.1007/s11356-022-19452-z
[100] Zulfiqar U, Haider F U, Ahmad M, et al. Chromium toxicity, speciation, and remediation strategies in soil-plant interface: A critical review[J]. Frontiers in Plant Science, 2023, 13: 1081624. doi: 10.3389/fpls.2022.1081624
[101] 张兆鑫, 曹宁宁, 李林记, 等. 原位吸附技术修复六价铬污染土壤[J]. 岩矿测试, 2024, 43(2): 302−314. doi: 10.15898/j.ykcs.202307090090 Zhang Z X, Cao N N, Li L J, et al. In situ adsorption technology for remediation of Cr(Ⅵ) contaminated soil[J]. Rock and Mineral Analysis, 2024, 43(2): 302−314. doi: 10.15898/j.ykcs.202307090090
[102] 杨梦楠, 孙晗, 曹海龙, 等. 生物炭-壳聚糖磁性复合吸附剂的制备及去除地下水中铅和铜[J]. 岩矿测试, 2023, 42(3): 563−575. doi: 10.15898/j.ykcs.202208230155 Yang M N, Sun H, Cao H L, et al. Preparation and application of biochar-chitosan magnetic composite adsorbent for removal of lead and copper from groundwater[J]. Rock and Mineral Analysis, 2023, 42(3): 563−575. doi: 10.15898/j.ykcs.202208230155
[103] Peng H, Gao P, Chu G, et al. Enhanced adsorption of Cu(Ⅱ) and Cd(Ⅱ) by phosphoric acid-modified biochars[J]. Environmental Pollution, 2017, 229: 846−853. doi: 10.1016/j.envpol.2017.07.004
[104] Mandal S, Sarkar B, Bolan N, et al. Enhancement of chromate reduction in soils by surface modified biochar[J]. Journal of Environmental Management, 2017, 186: 277−284. doi: 10.1016/j.jenvman.2016.05.034
[105] Murad H A, Ahmad M, Bundschuh J, et al. A remediation approach to chromium-contaminated water and soil using engineered biochar derived from peanut shell[J]. Environmental Research, 2022, 204: 112125. doi: 10.1016/j.envres.2021.112125
[106] Aparicio J D, Lacalle R G, Artetxe U, et al. Successful remediation of soils with mixed contamination of chromium and lindane: Integration of biological and physico-chemical strategies[J]. Environmental Research, 2021, 194: 110666. doi: 10.1016/j.envres.2020.110666
[107] Su H, Fang Z, Tsang P E, et al. Remediation of hexavalent chromium contaminated soil by biochar-supported zero-valent iron nanoparticles[J]. Journal of Hazardous Materials, 2016, 318: 533−540. doi: 10.1016/j.jhazmat.2016.07.039
[108] Ahmed T, Noman M, Ijaz M, et al. Current trends and future prospective in nanoremediation of heavy metals contaminated soils: A way forward towards sustainable agriculture[J]. Ecotoxicology and Environmental Safety, 2021, 227: 112888. doi: 10.1016/j.ecoenv.2021.112888
[109] Mondal P, Anweshan A, Purkait M K. Green synthesis and environmental application of iron-based nanomaterials and nanocomposite: A review[J]. Chemosphere, 2020, 259: 127509. doi: 10.1016/j.chemosphere.2020.127509
[110] Wei Y Z, Chu R, Zhang Q H, et al. Nano zero-valent iron loaded corn-straw biochar for efficient removal of hexavalent chromium: Remediation performance and interfacial chemical behaviour[J]. RSC Advances, 2022, 12(41): 26953−26965. doi: 10.1039/d2ra04650d
-
其他相关附件




 下载:
下载:

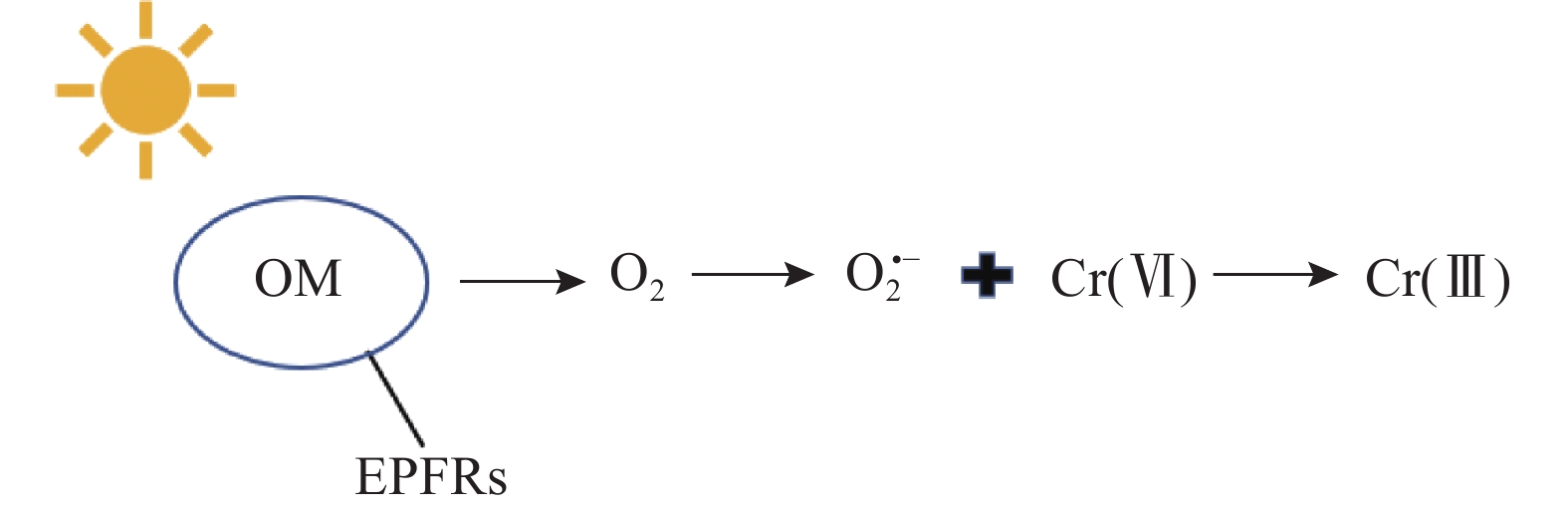
 京公网安备 11010202008159号
京公网安备 11010202008159号