Laser Induced Fluorescence for In Situ Detection of Typical Heavy Metals in Groundwater
-
摘要:
对地下水重金属污染进行现场检测,是快速识别重金属种类和评价污染程度的重要手段。激光诱导荧光技术(LIF)利用特定荧光探针,在目标重金属存在下,通过激光诱导荧光探针产生/猝灭荧光,从而完成对重金属的识别和检测,具有快速甄别地下水重金属污染特性和无损获取其价态的优势。本文在简述LIF检测重金属原理和LIF原位检测地下水重金属装备基础上,重点从荧光探针合成及其在重金属传感检测方法的构建上总结了基于不同荧光探针的LIF现场检测重金属的研究进展。目前可用于重金属检测的荧光探针包括以小分子探针、有机大分子和聚集诱导发光为主的有机荧光探针和以金纳米簇、量子点和金属有机框架材料为代表的纳米荧光探针。这些探针的合成及其对应的传感检测方法的构建表明LIF技术在地下水重金属检测中具有巨大优势。而针对地下水重金属检测的LIF设备研发方面的成果虽不如荧光传感检测方法丰富,但已研制出部分重金属的传感器和检测装备,表现出良好的应用前景。未来研究将会聚焦在地下水重金属主控因子识别和抗干扰技术研发、荧光探针合成与LIF检测方法构建、重金属传感部件和检测装备集成研制,以及检测技术标准化,为后续场地地下水典型重金属LIF检测技术的研究提供参考。
要点(1)基于LIF的地下水重金属检测不能直接通过LIF检测重金属,而是通过荧光探针与地下水中重金属进行特异性结合,使得探针原有荧光信号发生改变,从而间接指示或检测出目标重金属。
(2) LIF荧光探针主要为有机分子探针和纳米探针,有机分子探针通常本身携带有羰基、羟基或羧基等基团能特异性识别重金属,展现出良好的特异性,而大部分纳米探针没有识别基团,会辅以适配体等识别材料增强探针特异性。
(3) LIF检测地下水重金属研究未来将聚焦于地下水抗干扰技术研发、LIF敏感材料合成与检测方法构建、LIF集成装备研制及检测标准化等方面。
HIGHLIGHTS(1) LIF detects heavy metals through specific binding of fluorescent probes with heavy metals, causing probe signal changes and indirectly indicating or detecting targets.
(2) LIF probes consist of organic probes and nanoprobes. The former usually carry functional groups such as carbonyl, hydroxyl, or carboxyl groups, which can specifically recognize metals and exhibit good specificity, while the latter without recognition groups are supplemented with recognition elements such as aptamer to enhance specificity.
(3) LIF for heavy metals in groundwater will focus on development of anti-interference technique, synthesis of sensitive materials and corresponding methods construction, integration of equipment, and detection standardization.
Abstract:On-site detection of heavy metals in groundwater is important to quickly evaluate pollution. Laser induced fluorescence (LIF) utilizes specific fluorescent probes to generate/quench fluorescence in the presence of heavy metals, thereby achieving heavy metals detection, which can quickly identify heavy metals and non-destructively obtain their valence states. This work summarizes the principle and equipment of, and its application for, in situ detection of typical heavy metals in groundwater. At present, LIF probes used for heavy metal detection include organic fluorescent probes mainly composed of small molecule probes, macromolecules, and AIE probes, as well as nanomaterial probes represented by gold nanoclusters, QDs, and MOFs. These synthesized probes and the corresponding constructed sensors indicate that LIF holds significant advantages in heavy metal detection in groundwater. Although the achievements in the development of LIF equipment for heavy metal detection in groundwater are not as rich as those in sensing methods, LIF equipment for several heavy metals have been developed, demonstrating good application prospects. Future research will focus on identifying the controlling factors of heavy metals in groundwater and developing anti-interference techniques, synthesizing novel fluorescent probes for LIF sensor, integrating sensing components into LIF equipment, and standardizing the LIF detection process. The BRIEF REPORT is available for this paper at http://www.ykcs.ac.cn/en/article/doi/10.15898/j.ykcs.202402230018.
-
Keywords:
- groundwater /
- heavy metals /
- laser induced fluorescence /
- fluorescent probe /
- on-site detection
BRIEF REPORTWith the rapid development of non-ferrous metal mining, smelting, and electroplating, heavy metals such as mercury (Hg), cadmium (Cd), arsenic (As), lead (Pb), and chromium (Cr) enter the groundwater through various pathways[1-3], which are not only non-degradable, causing continuous pollution of groundwater, but also heavy metals that remain in groundwater can enter the human body through the enrichment and amplification of the food chain, posing a threat to human health[6]. Therefore, establishing effective analytical methods is crucial for identifying, and evaluating heavy metal pollution in groundwater on site. The laser induced fluorescence (LIF) technique requires only a constant current driven small package laser diode or light-emitting diode at the hundred-milliwatt level to excite fluorescence probes and obtain their fluorescence spectra, which allows for non-destructive analysis of heavy metals, providing the possibility of detecting low concentrations of heavy metals in groundwater[10-12].
The LIF principle is to let a beam of laser pass through the detection area, adjust the laser wavelength, and when the energy of the laser photon (related to the wavelength) is equal to the energy difference between two specific energy levels of a target in the detection area, the target absorbs the photon energy and transitions to a high-energy state[15]. The target molecules in high-energy states are unstable and return to the ground state within a certain period of time. During this process, target molecules emit energy and fluorescence through spontaneous emission[16-17]. Due to the low intrinsic fluorescence efficiency of heavy metals, LIF has difficulty in directly detecting heavy metals unlike XRF. Instead, various fluorescent probes must be supplemented with specific binding of heavy metals in the sample to change the original fluorescence probe signal, thereby indirectly indicating or detecting the targets[20],such as the cyanine fluorescence probe for methylmercury[21].
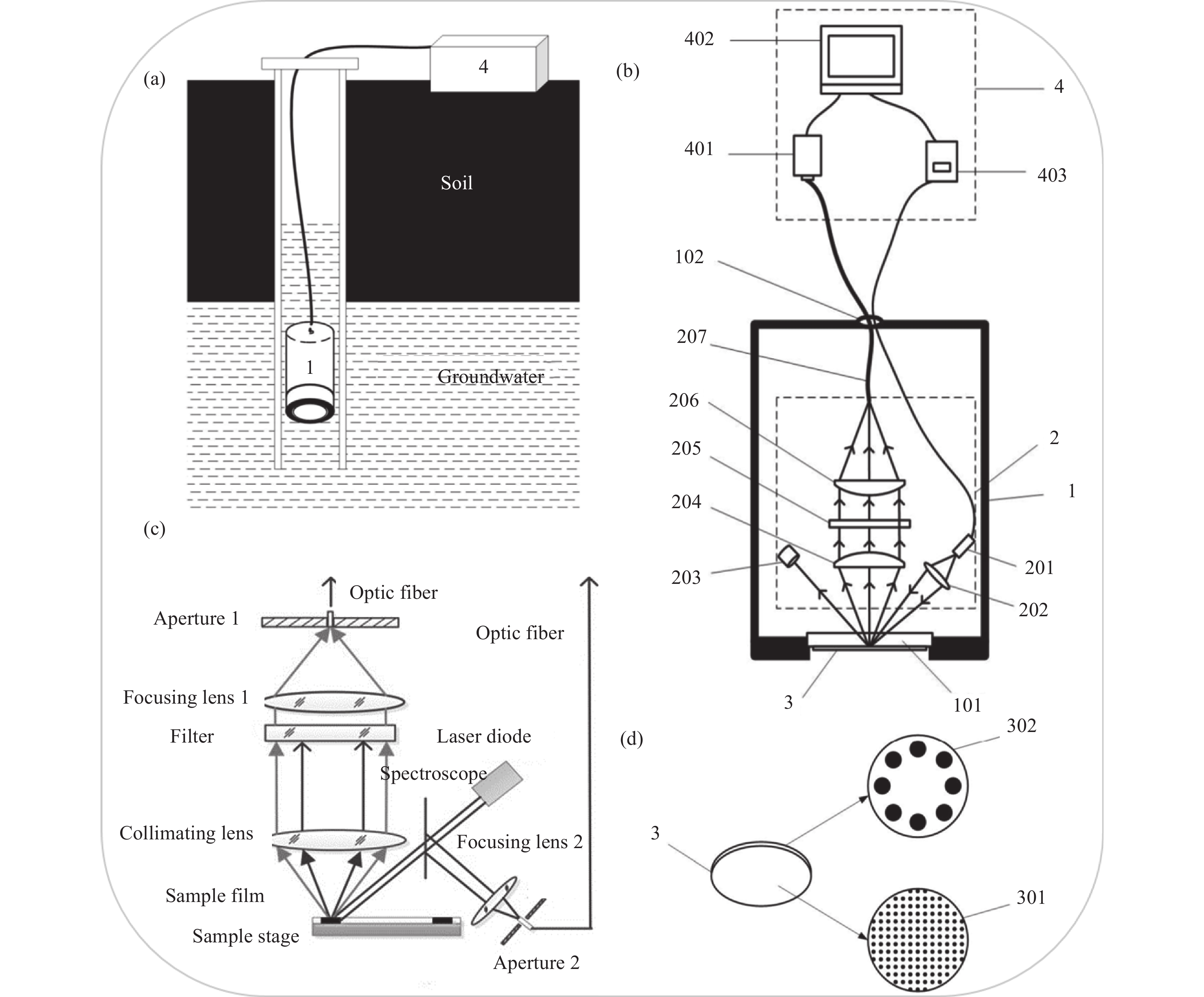
The LIF equipment for heavy metals in groundwater mainly comprises a laser, optical components, sample cell, photodetector, signal processing module, and related accessory components (Fig.1)[22]. Given the complexity and requirements of on-site heavy metals detection in groundwater, LIF equipment needs to be designed and optimized in terms of light source, constant temperature system, optical path, as well as noise reduction, sensitivity enhancement, and stability faced by on-site detection[23-24]. Three specific aspects need to be considered: (1) the diameter of the groundwater monitoring well head is only 110mm, requiring portable detection equipment and miniaturization of detection probes; (2) the heavy metal concentration in groundwater is low, requiring a high excitation efficiency of the laser light source; (3) it is necessary to hold good equipment stability due to the ever-changing and complicated environment in the field.
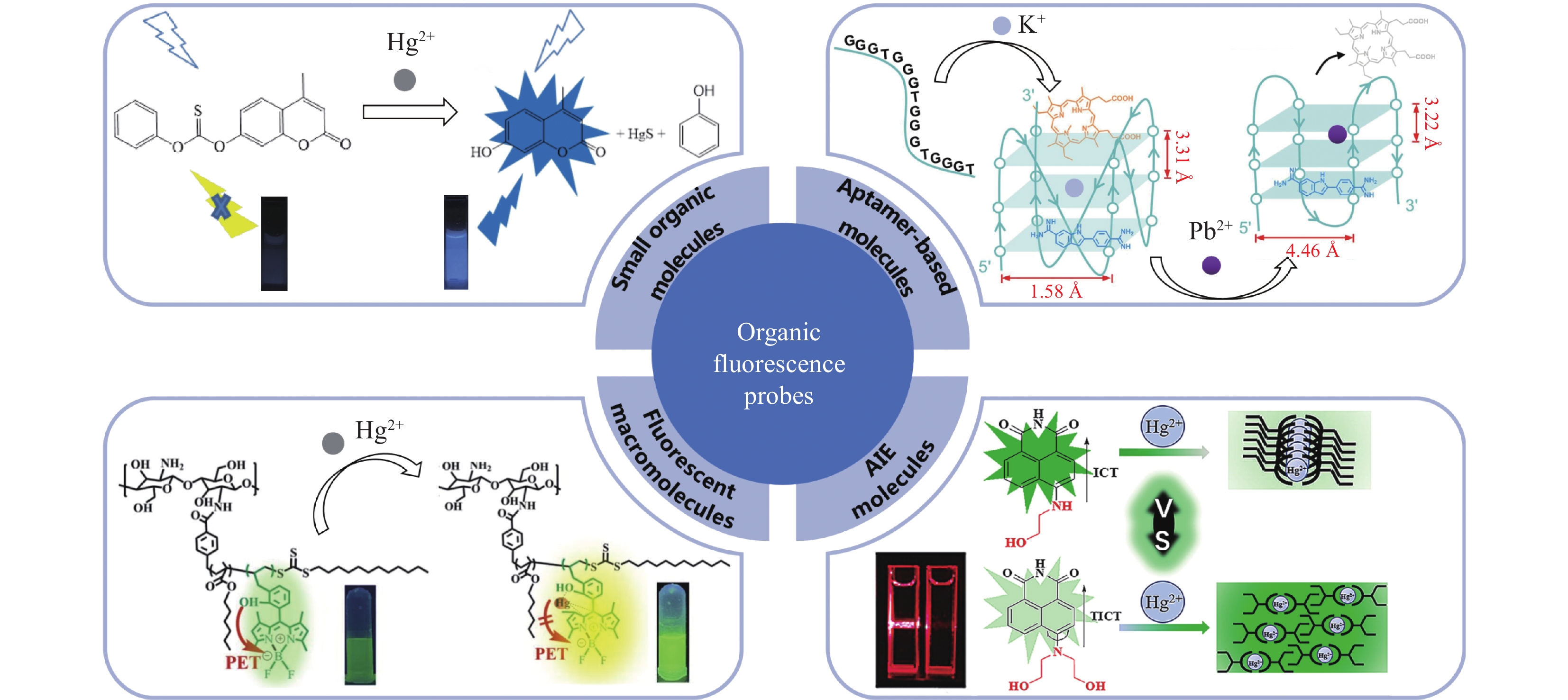
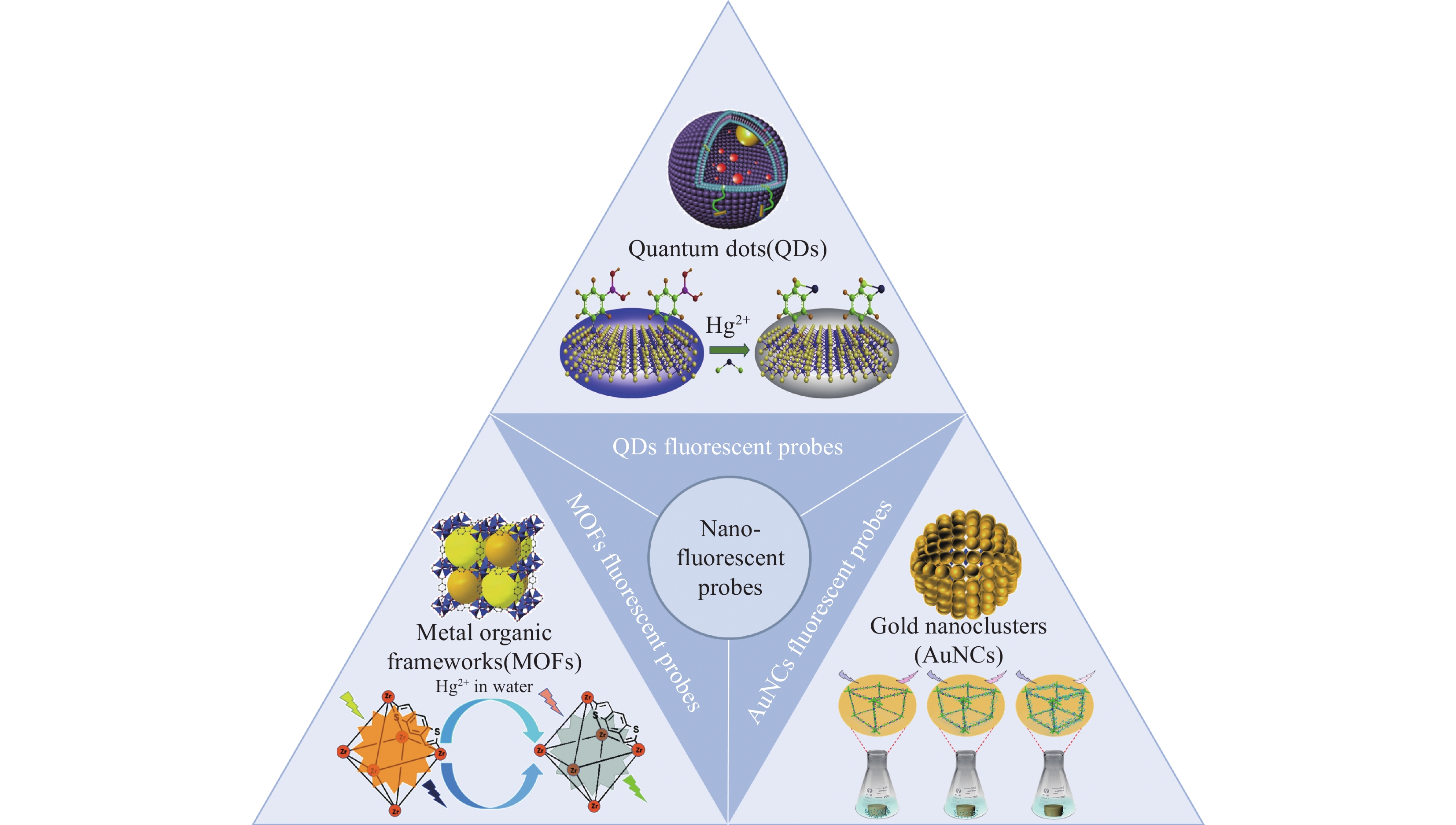
LIF probes for heavy metals mainly include organic and nanomaterial fluorescent probes. Organic fluorescent probes hold the advantage of a variety of types, tunable structures, high fluorescence quantum efficiency, and tunable spectra, and they mainly comprise organic small molecule probes, aggregation-induced emission (AIE) probes, and macromolecule probes (Fig.2). Nanomaterial fluorescent probes have advantages of high fluorescence quantum efficiency and good fluorescence stability, mainly including gold nanoclusters, quantum dots, and metal-organic framework probes (Fig.3). These fluorescent probes have been applied in the detection of typical heavy metal ions such as Hg, Cd, As, Pb, and Cr, and have shown good detection sensitivity and specificity under laboratory conditions. Table 1 shows that many fluorescent probes can specifically recognize certain heavy metal ions to a certain extent due to carrying carbonyl, hydroxyl, or carboxyl groups, such as coumarin probes that recognize Hg by thiocarbonate. However, some probes that do not have specific recognition groups, such as gold nanoclusters, and due to the complex composition of groundwater and the significant influence of environmental factors, it is necessary to further enhance the specificity of LIF probes. Aptamers can bind with targets with high affinity and strong specificity, making them a promising heavy metal recognition element for LIF detection. Apart from Cr, the other heavy metals, including As, Hg, Pb, and Cd, have their own aptamers, which to some extent compensates for insufficient specificity of LIF probes.
Future studies should focus on the following aspects: identifying the main influencing factors of heavy metals in groundwater and developing anti-interference techniques, synthesizing fluorescent probes and constructing LIF methods, integrating sensing components and LIF equipment, and standardizing the detection process. (1) It is necessary to investigate the groundwater composition, heavy metal concentration and forms, and solid-liquid distribution patterns, combined with the geological and hydrogeological conditions in groundwater, to identify the distribution characteristics of heavy metals at the site scale, with a focus on identifying the hydrological and environmental biogeochemical coupling processes and main influencing factors that affect the distribution and migration of metal forms. (2) It is necessary to consider the detection of low concentration and complex forms of heavy metal ions in groundwater from polluted sites as the goal, and to accurately synthesize high sensitivity and high selectivity sensitive materials with fluorescence signals. A precise synthesis system for materials sensitive to typical heavy metals with fluorescence response signals should be constructed. We should select novel LIF probes with high fluorescence efficiency, good stability, and strong specificity, and evaluate analytical performance. Then, in order to achieve the goal of detecting low concentrations of heavy metals, LIF methods for identifying heavy metals in specific scenarios of groundwater should be established. (3) The developed anti-interference techniques and LIF probes would be utilized as components to study adaptive noise reduction filtering and efficient extraction for LIF signals, making them suitable for heavy metals in groundwater. In situ sensing optical paths, chemical modifications, and integrated process structures would be designed, and precise LIF equipment for heavy metals in groundwater would be developed. A multi-channel parallel sensing signal processing and analysis system should be designed and be highly embedded and integrated with various functional module units, to achieve device miniaturization and portability; it should optimize data processing, and establish typical heavy metal detection models; building an IoT cloud platform is necessary to form an in situ detection equipment that integrates site collection, transmission, and management. (4) Typical demonstration sites in various regions and industries should be selected for LIF equipment validation, and feedback optimization should be carried out in continuous verifications in groundwater. Meanwhile, by comprehensively utilizing detailed validation and analysis data from the demonstration site and groundwater data from the national environmental monitoring network, advanced methods such as machine learning should be developed to explore adaptive environmental factor compensation and correction techniques, and ultimately achieve standardization of LIF-based detection.
Substantial progress has been achieved in LIF for heavy metals in groundwater. LIF for in situ detection of heavy metals in groundwater has mainly focused on the synthesis of organic and nanomaterial probes targeting heavy metals, as well as the construction of corresponding LIF methods, which indicates that LIF holds significant advantages in on-site detection of heavy metals in groundwater. Although the achievements in the development of LIF equipment for heavy metal detection in groundwater are not as rich as those in fluorescence sensing methods, corresponding sensors and equipment have been developed for several typical heavy metals, demonstrating good application prospects. Although numerous fluorescent probes have been applied to heavy metal detection, LIF is still in its early stages in detecting typical heavy metals in groundwater. Most existing fluorescent probes have been preliminarily explored in laboratory conditions and have not yet been widely applied to actual groundwater detection. The developed LIF equipment also lacks sufficient anti-interference or stability, making it difficult to meet the detection of complex groundwater in different scenarios. Herein, LIF for heavy metals in groundwater needs further development in anti-interference technique, fluorescent probes, and sensor equipment and detection process standardization.
-
土壤矿物中能够被作物直接吸收利用的部分,即有效量,以活动态存在于土壤中[1-2]。元素对环境或生物体产生的效应或毒性,在很大程度上取决于其各形态含量,而不是总浓度[3-4]。土壤微量元素有效量不但影响作物的生长发育,而且影响作物品质和产量[5-8]。因此元素的形态、有效态分析越来越受到研究人员的重视[9-10]。通过土壤有效磷的测定,有助于了解土壤供应磷的情况,为合理使用磷肥及提高磷肥利用率提供依据[11-13]。通常所谓的土壤有效磷是指某一特定形态的磷,只是一个相对指标,但是其可用于说明土壤的供磷水平[14-15]。
目前,测定土壤有效磷的方法主要有Olsen法和Bray法[16]。Olsen法用碳酸氢钠溶液浸提土壤,Bray法用氟化铵-盐酸溶液浸提土壤。方法原理是浸提土壤后,分取浸提液加入显色剂,使用分光光度法测定浸提液中的磷含量。其中Bray法对酸性土壤效果良好,Olsen法对中性及石灰性土壤效果良好[17]。现行标准方法《土壤检测第7部分土壤有效磷的测定》(NY/T 1121.7—2014)和《森林土壤磷的测定》(LY/T 1232—2015)中Olsen法和Bray法的应用存在参数差异[18],主要差异在于酸性土壤的浸提时间、振荡温度等。目前常用的土壤有效态标准样品中,ASA1-6(GBW07412)、NSA1-6是依据GB/T 7853—1987定值,ASA1a至ASA6a、ASA-7至ASA-10是依据NY/T 1121.7—2006定值(GB/T 7853—1987、NY/T 1121.7—2006分别已被LY/T 1232—2015、NY/T 1121.7—2014替代)。已有文献报道了使用电感耦合等离子体发射光谱法(ICP-OES)测定浸提液中的有效磷[19-22],由于ICP-OES测定磷的灵敏度低,检出限高,达不到部分土壤样品的有效磷含量,因此分光光度法仍是测定土壤中有效磷的最优选择。该技术存在以下问题:①现有测定方法需浸提后过滤得到澄清待测液,过滤操作繁琐,且有些土壤沉淀颗粒细,极易穿透滤纸,速度慢效率低。②浸提液显色前必须先将pH调至指定值,再加钼锑抗显色剂才能发生特定显色反应。且该方法常使用二硝基酚(变色范围为无色pH 2.4~4.0黄色)作指示剂,由黄色调至恰变浅黄,而土壤浸提液多略带黄色的底色,尤其是有机质含量高的土壤,浸提滤液易呈现黄或褐色[23-25],导致调pH时颜色无法判断;且二硝基酚属于易制爆试剂,毒性强[26]。
针对以上问题,本文采用离心分离代替过滤;比较了不同浸提时间、不同浸提液酸度、不同酸碱指示剂等实验条件对检测结果的影响;使用溴酚蓝作指示剂调节pH,对比了溴酚蓝指示剂与标准方法的二硝基酚指示剂的检测结果。拟通过对现有方法进行优化创新,提高检测效率。
1. 实验部分
1.1 仪器及工作条件
分光光度计(北京普析通用仪器有限责任公司,型号T9CS),离心机(天津广丰科技有限公司,型号TD6M),恒温振荡培养箱(苏州培英实验设备有限公司,型号HZQ-X100)。
1.2 材料和主要试剂
磷标准溶液1000mg/L(国家有色金属及电子材料分析测试中心)。
溴酚蓝指示剂:称取0.20g溴酚蓝溶于100mL乙醇中。
盐酸(北京化学试剂研究所,BⅧ级);盐酸(国药集团化学试剂有限公司,光谱纯);氟化铵(Strem Chemicals,ACS);氟化铵(上海麦克林生化科技有限公司,ACS)。
酸性土壤:土壤有效态成分分析标准样品GBW07416、GBW07412(中国地质科学院地球物理地球化学勘查研究所);未知样A(野外采集,测定pH < 6.5)。
中性及石灰性土壤:土壤有效态成分分析标准样品GBW07413、GBW07414、GBW07414a(中国地质科学院地球物理地球化学勘查研究所);未知样B(野外采集,测定pH>6.5)。
其他试剂参考标准方法《土壤检测第7部分土壤有效磷的测定》(NY/T 1121.7—2014)配制,均采用市售分析纯。
1.3 实验条件
参考标准方法《土壤检测第7部分土壤有效磷的测定》(NY/T 1121.7—2014)。
酸性土壤:①待测液的制备:称取酸性土壤5.00g于200mL塑料浸提瓶中,加氟化铵(0.03mol/L)-盐酸(0.025mol/L)浸提液50.0mL,25℃以160r/min恒温振荡60min后,5000r/min离心5min。②移取上清液10mL于50mL比色管中,加入5.00mL硼酸(30g/L),加水,加入溴酚蓝指示剂2滴,用2mol/L盐酸和2mol/L氨水调节至试液刚呈黄色。③测定:加5.00mL钼锑抗显色剂,定容至50mL,摇匀,25℃以上放置30min后用分光光度计在700nm或880nm处进行吸光度测定,绘制标准曲线,计算出待测样品中有效磷含量。
中性或石灰性土壤:①待测液的制备:称取中性或石灰性土壤2.50g于200mL塑料浸提瓶中,加50.0mL碳酸氢钠浸提液(42g/L),25℃以160r/min恒温振荡30min后,5000r/min离心5min。②移取上清液10mL于50mL比色管中,加入溴酚蓝指示剂2滴,用0.5mol/L硫酸调节至试液刚呈黄色。③测定:加5.00mL钼锑抗显色剂,定容至50mL,摇匀,25℃以上放置30min后用分光光度计在700nm或880nm处进行吸光度测定,绘制标准曲线,计算出待测样品中有效磷含量。
2. 结果与讨论
2.1 酸性土壤浸提时间及土水比实验结果
用土壤有效态国家标准物质GBW07416开展条件实验,改变酸性土壤浸提时间及水土比,其他条件参考标准方法《土壤检测第7部分土壤有效磷的测定》(NY/T 1121.7—2014)。结果如图 1所示,氟化铵-盐酸偏提取酸性土壤时,随着浸提液比例降低,测定结果降低;随着提取时间增加,提取出的有效磷逐渐减少,与文献报道结果一致。王荣辉等[18]提出,可能是由于土壤体系自身缓冲作用,浸提时间延长,土壤体系pH升高,导致F-对Fe3+和Al3+的配合能力减弱,Fe-P和Al-P的释放量减少而引起。用GBW07416标准物质研究氟化铵-盐酸浸提时间与pH关系,结果如图 2所示,与文献[18]结论一致,且与不同浸提时间的测定结果一致。
本实验中使用标准方法的土水比1:10时,需浸提60min,才得到标样值结果。而中性及石灰性土壤可依据标准方法的实验条件测得相应的标样值。
2.2 溴酚蓝指示剂的对比实验
配制标准曲线0、1、5、10、20、30μg/50mL点,并选用国家标准物质GBW07412、GBW07414a、未知土壤样品A、未知土壤样品B及流程空白溶液,调节浸提液pH时分别使用二硝基酚和溴酚蓝作指示剂,用1cm比色池,比较两种方法的吸光度和检测结果。通过比较酸性土壤和中性、石灰性土壤标准曲线点的吸光度,结果表明用这两种指示剂调节pH后,加入显色剂,显色吸光度没有明显差异。表 1是分别用酸性土壤方法、中性和石灰性土壤方法分析对应的土壤未知样、标准物质、空白对比实验结果,可见使用溴酚蓝替代二硝基酚,检测结果完全一致,标准物质结果符合要求,说明本文优化方法可以适用于酸性、中性和石灰性土壤。使用溴酚蓝(变色范围黄色pH 3.0~4.6蓝色)作指示剂调至适宜pH值时颜色为黄色,磷酸根与钼锑抗显色剂形成蓝色配合物,两种颜色波长差异非常大,不干扰后续显色的测定。而且钼锑抗显色剂呈强酸性,加入后不会使待测液pH值向碱性变化而导致溴酚蓝显色剂变色影响实验结果。
表 1 使用不同pH指示剂测定土壤中有效磷结果对比Table 1. Comparison of analytical results of available phosphorus in soil extracted by different pH indicator不同酸碱性土壤 待测液调节pH指示剂 实验样品 吸光度 溶液中有效磷浓度
(μg/50mL)样品中有效磷测定值
(μg/g)有效磷标准值
(μg/g)酸性土壤 二硝基酚 GBW07412 0.245 22.305 22.30 21.2±3.6 未知样A 0.099 8.568 8.57 / 空白溶液 0.009 0.153 / / 溴酚蓝 GBW07412 0.266 23.648 23.65 21.2±3.6 未知样A 0.104 8.458 8.46 / BK 0.006 -0.070 / / 中性或石灰性土壤 二硝基酚 GBW07414a 0.140 14.721 29.44 29±3 未知样B 0.049 6.286 12.57 / 空白溶液 -0.017 0.263 / / 溴酚蓝 GBW07414a 0.116 14.248 28.50 29±3 未知样B 0.024 5.790 11.58 / 空白溶液 -0.020 0.013 / / 由于土壤样品的复杂性,偏提取得到的待测溶液可能含有未知物质,由于偏提取过程中化学反应的复杂性,土壤中的未知物有可能会干扰所用指示剂溴酚蓝的使用效果,但现有方法所用指示剂二硝基酚也存在这种未知性,本方法应用中尚未见不适用情况。
2.3 浸提液pH的影响
标准方法《土壤检测第7部分土壤有效磷的测定》(NY/T 1121.7—2014)中碳酸氢钠浸提液配制后,需用氢氧化钠溶液调节pH至8.5,并定期监测浸提液pH值。选用两个标准物质,配制后直接使用并与调节pH后的浸提液分别测定,有效磷结果见表 2,碳酸氢钠浸提液pH值对结果影响很大,配制后溶液pH为8.22,其浸提检测结果明显偏低。
表 2 碳酸氢钠浸提液pH对有效磷检测结果的影响Table 2. Effect of NaHCO3 extraction with different pH on analytical results of available phosphorus标准物质编号 有效磷测定值(μg/g) 有效磷标准值
(μg/g)未调节pH 调节pH至8.5 GBW07413 14.20 17.49 18.3±2 GBW07414 10.26 12.71 13.8±2.3 氟化铵-盐酸浸提液的pH在标准方法中未作强调,部分文献[27]中偶见pH 1.5~2.0。如2.1章节所述,由于本研究中的浸提时间与行业标准方法《土壤检测第7部分土壤有效磷的测定》(NY/T 1121.7—2014)不一致,且碳酸氢钠碱性浸提液pH对实验结果有主要影响,为了了解酸性浸提液pH的影响程度,实验中尝试用不同厂家的氟化铵及盐酸试剂,按标准方法配制的酸性浸提液pH值为2.7~3.2,测定结果没有明显差异。实验表明将氟化铵-盐酸浸提液pH调节至2后,浸提出大量的磷,明显不适用于有效磷的测定,因此氟化铵-盐酸浸提液配制后可直接使用。实验中发现,氟化铵-盐酸浸提液放置一个月,pH值由2.72略升高至2.79,其浸提GBW07416一小时后溶液pH值为5.04,也比现配的溶液浸提后的pH值(4.90)高。虽检测结果未见明显差异,但浸提液的pH值可能会影响浸提能力,最好每周配制。
以上实验表明,碳酸氢钠浸提液配制后必须调节pH至8.5,氟化铵-盐酸浸提液配制后可直接使用。
2.4 已浸提溶液的存放时间
《森林土壤有效磷的测定》(LY/T 1233—1999)提到浸提后溶液的保存时间,需24h之内测定。本研究测定了两个不同含量水平的酸性土壤样品,结果见表 3,有效磷含量在2μg/g左右的样品,浸提液滤液放置3天检测结果下降约7.57%,放置5天检测结果下降约9.17%;有效磷含量在15μg/g左右的标准样品(GBW07416),浸提液滤液放置3天检测结果下降约0.43%,放置5天检测结果下降约13.20%。可见浸提液久置后有效磷会损失,其含量下降程度与样品及含量有关,最好在浸提当天完成检测。
表 3 已浸提溶液的存放时间对有效磷检测结果的影响Table 3. Effect of extracted solution with different storage time on analytical results of available phosphorus样品 放置不同时间有效磷测定值(μg/g) 有效磷测定值下降比例(%) 0d 3d 5d 3d 5d 未知样品 2.54 2.34 2.30 -7.57 -9.17 GBW07416 17.05 16.98 14.80 -0.43 -13.20 2.5 方法检出限和精密度
选用酸性土壤有效态标准物质GBW07416和碱性土壤有效态标准物质GBW07414,分别平行偏提取7份,检测结果见表 4。GBW07416检测结果平均值为17.2μg/g,GBW07414检测结果平均值为12.5μg/g,均在标准值范围内,准确度和相对标准偏差符合要求。依据《环境监测分析方法标准制修订技术导则》(HJ 168—2010)附录A.1.2,使用3cm比色皿时,与0.01吸光度对应的浓度值作为检出限,对应曲线浓度点约为0.5μg/50mL。称样量为5.0g分取10mL浸提液时,检出限为0.5μg/g;称样量为2.5g分取10mL浸提液时,检出限为1.0μg/g。
表 4 方法精密度Table 4. Precision tests of the method参数 GBW07416 GBW07414 有效磷测定平均值(μg/g) 17.2 12.5 有效磷标准值(μg/g) 14.8±3.1 13.8±2.3 标准偏差(n=7) 0.5907 0.2323 相对标准偏差(%,n=7) 3.44 1.86 3. 结论
本研究探讨了测定土壤中有效磷标准方法中的实验条件对检测结果的影响,以及酸性土壤浸提时间及土水比对测定结果的影响。结果表明,酸性土壤在1:10土水比条件下,需浸提60min可得到标样值结果,而碱性土壤可依据标准方法实验条件得到标样值结果;同时探讨了浸提液pH的影响,碳酸氢钠浸提液需调pH至8.5,否则结果明显偏低,氟化铵-盐酸浸提液不可调pH且不可久置;对于已浸提溶液的存放时间,最好在浸提当天完成检测。
本研究采用溴酚蓝指示剂替代标准方法的二硝基酚指示剂,标准方法使用的二硝基酚指示剂颜色突变不明显,易受浸提液底色影响,且易制爆、毒性强;而溴酚蓝指示剂调节pH时突变明显,不受浸提液底色干扰,优于标准方法的二硝基酚指示剂。基于两种指示剂的吸光度和检测结果完全一致,表明溴酚蓝指示剂可适用于土壤中有效磷的测定。
-
图 1 LIF地下水重金属原位检测装置(a)外部结构示意图,(b)内部结构示意图,(c)光路图以及(d)修饰有选择性敏感材料的敏感膜结构示意图[22]
1—防水腔体,101—通光窗口,102—光纤与信号线防水接口;2—光学系统,201—激光二极管,202—激发光准直聚焦镜组,203—激发光光陷阱,204—荧光准直镜,205—滤光片,206—荧光聚焦镜,207—光纤;3—重金属膜,301—拦截膜,302—响应膜;4—信号处理与控制单元,401—光电探测器,402—控制中心,403—光源驱动模块。
Figure 1. Schematic diagram of (a) the external structure, (b) the internal structure, (c) optical path, and (d) membrane structure modified with selective and sensitive materials of LIF device for on-site detection of heavy metal in groundwater [22]
表 1 荧光探针检测重金属离子(有机荧光探针和纳米荧光探针)
Table 1 Fluorescent probes (organic and nanomaterial fluorescent probes) for the detection of heavy metals
荧光探针
材料类型荧光探针 识别元件 靶标 检出限 线性范围 样品 优点 缺点 参考
文献有机
荧光探针
材料罗丹明衍生物 螺旋内酰胺 Pb2+ 0.56μg/L 0.21~210μg/L 海产品 可视化,检测范围宽 合成复杂 [25] 喹啉类 羰基 Cd2+ 132μg/L / 活细胞 荧光稳定 低灵敏度 [26] 香豆素类 硫代碳酸盐 Hg2+ 1678.3μg/L / 河水 高特异性 低灵敏度 [27] 适配体基 三螺旋核酸分子 As(Ⅲ) 5ng/L 10ng/L~10mg/L 湖水 高特异性,高灵敏度和
检测范围宽修饰复杂 [28] Pb2+适配体 Pb2+ 12.22μg/L 20.7~518.0μg/L 茶叶 高特异性,合成简单 成本较高 [29] AIE分子 萘酰亚胺 Hg2+ 4.35μg/L 20.7~103.6μg/L 湖水 重复性好,特异性强 线性范围窄 [30] 偶氮四唑 Hg2+ 0.394μg/L 79.665μg/L~2.072mg/L 地下水 荧光稳定,特异性强 抗干扰能力弱 [31] 改性壳聚糖 氨基、羟基 Hg2+/Hg+ 126.4/97.4μg/L / / 有机可溶,抗干扰能力和
高重复性好合成复杂,成本高 [32] 纳米
荧光探针
材料碳量子点 羟基、羧基 Cr6+ 3.7mg/L 5~200mg/L 井水和湖水 环境友好 低灵敏度,线性范围窄 [33] Cu@AuNCs 胸腺嘧啶 Hg2+ 1.385μg/L 140.8~1971.3μg/L 水产品 特异性强,稳定性好 / [34] AuNCs 胸腺嘧啶 Hg2+ 0.59μg/L 2.8~84.486μg/L 河水和湖水 高灵敏度,高选择性 线性范围窄 [35] UCNPs-AuNCs UCNPs Pb2+ 0.216μg/L 0~2.63μg/L 污水 高选择性(可检测复合
金属离子中的Pb2+)/ [36] AuNCs 适配体 Pb2+ 14.99ng/L 26.3~52595.8 ng/L 自来水 高灵敏度 检测时间长 [37] N-GQDs 羟基 Fe3+ 72.99μg/L 0~81102μg/L 水溶液 量子产率高,检测时间短 灵敏度低 [38] CQDs-AgNPs AgNPs Hg2+ 0.02816μg/L 0.14081~140.810μg/L 湖水和废水 量子产率高,重复性好 / [39] ZnSe QDs 四乙氧基硅烷 Pb2+ 0.335μg/L 1~60μg/L 海水和湖水 重复性好,多通道检测 合成复杂 [40] N-CQDs 羟基、羧基 Pb2+ 0.507μg/L 0~10.52μg/L 自来水和池塘水 高灵敏度,抗干扰能力强 / [41] ZIF-8 适配体 Cd2+ 0.018ng/L 0.02364~28.37ng/L 自来水 超高灵敏度 / [42] 2D-MOF 适配体 Pb2+ 0.174μg/L 0.263~52.6μg/L 自来水 高选择性,检测时间短 / [43] ZnMOF-74 酚羟基 Fe3+ 2.22μg/L 5.55~5554.34μg/L 河水 高选择性 灵敏度低 [44] Eu3+@UIO-66 2,6-二羧基吡啶 Hg2+ 2.326μg/L 2.816~70.4μg/L 水溶液 稳定性好,特异性强 / [45] -
[1] 杨梦楠, 孙晗, 曹海龙, 等. 生物炭-壳聚糖磁性复合吸附剂的制备及去除地下水中铅和铜[J]. 岩矿测试, 2023, 42(3): 563−575. doi: 10.15898/j.ykcs.202208230155 Yang M N, Sun H, Cao H L, et al. Preparation and application of biochar-chitosan magnetic composite adsorbent for removal of lead and copper from groundwater[J]. Rock and Mineral Analysis, 2023, 42(3): 563−575. doi: 10.15898/j.ykcs.202208230155
[2] 刘斯文, 黄园英, 赵文博, 等. 赣南北部黄陂河流域离子型稀土矿地区水质与健康风险评价[J]. 岩矿测试, 2022, 41(3): 488−498. doi: 10.15898/j.cnki.112131/td.202111080170 Liu S W, Huang Y Y, Zhao W B, et al. Water quality and health risk assessment of ion type rare earth deposits in the Huangpi River Basin of Northern Jiangxi Province[J]. Rock and Mineral Analysis, 2022, 41(3): 488−498. doi: 10.15898/j.cnki.112131/td.202111080170
[3] Huang C, Guo Z, Li T, et al. Source identification and migration fate of metal(loid)s in soil and groundwater from an abandoned Pb/Zn mine[J]. Science of the Total Environment, 2023, 895: 165037. doi: 10.1016/j.scitotenv.2023.165037
[4] Xu M, Zhang K, Wang Y, et al. Health risk assessments and microbial community analyses of groundwater from a heavy metal-contaminated site in Hezhou City, Southwest China[J]. International Journal of Environmental Research and Public Health, 2023, 20(1): 604. doi: 10.3390/ijerph20010604
[5] 钟林健. 铅锌矿区地下水位对尾矿中重金属在土壤中吸附与迁移的影响研究[D]. 长沙: 中南大学, 2023. Zhong L J. Study on the influence of groundwater level on the adsorption and migration of heavy metals from tailings in soil in lead-zinc mining area[D]. Changsha: Central South University, 2023.
[6] Lei K, Giubilato E, Critto A, et al. Contamination and human health risk of lead in soils around lead/zinc smelting areas in China[J]. Environmental Science and Pollution Research, 2016, 23: 13128−13136. doi: 10.1007/s11356-016-6473-z
[7] 李谨丞, 曹文庚, 潘登, 等. 黄河冲积扇平原浅层地下水中氮循环对砷迁移富集的影响[J]. 岩矿测试, 2022, 41(1): 120−132. doi: 10.15898/j.cnki.11-2131/td.202110080140 Li J C, Cao W G, Pan D, et al. The impact of nitrogen cycling on arsenic migration and enrichment in shallow groundwater of the Yellow River alluvial fan plain[J]. Rock and Mineral Analysis, 2022, 41(1): 120−132. doi: 10.15898/j.cnki.11-2131/td.202110080140
[8] 周兴辉, 胡敬芳, 宋钰, 等. 便携式水质重金属电化学检测仪的研究进展[J]. 传感器世界, 2020, 26(3): 7−13. doi: 10.16204/j.cnki.sw.2020.03.001 Zhou X H, Hu J F, Song Y, et al. Research progress on portable electrochemical detection instruments for heavy metals in water quality[J]. Sensor World, 2020, 26(3): 7−13. doi: 10.16204/j.cnki.sw.2020.03.001
[9] Yu L, Pang Y, Mo Z, et al. Coordination array for accurate colorimetric sensing of multiple heavy metal ions[J]. Talanta, 2021, 231: 122357. doi: 10.1016/j.talanta.2021.122357
[10] Meng D, Zhao N, Wang Y, et al. On-line/on-site analysis of heavy metals in water and soils by laser induced breakdown spectroscopy[J]. Spectrochimica Acta Part B: Atomic Spectroscopy, 2017, 137: 39−45. doi: 10.1016/j.sab.2017.09.011
[11] Yang Z, Ren J, Du M, et al. Enhanced laser-induced breakdown spectroscopy for heavy metal detection in agriculture: A review[J]. Sensors, 2022, 22(15): 5679. doi: 10.3390/s22155679
[12] 孔维恒, 曾令伟, 饶宇, 等. 基于预分类策略的激光诱导击穿光谱技术用于岩石样品定量分析[J]. 岩矿测试, 2023, 42(4): 760−770. doi: 10.15898/j.ykcs.202212190234 Kong W H, Zeng L W, Rao Y, et al. Laser induced breakdown spectroscopy technology based on pre classification strategy for quantitative analysis of rock samples[J]. Rock and Mineral Analysis, 2023, 42(4): 760−770. doi: 10.15898/j.ykcs.202212190234
[13] 文志明. 荧光光度法及其在环境分析化学中的应用[J]. 中国环境监测, 1992, 8(2): 45−55. doi: 10.19316/j.issn.1002-6002.1992.02.018 Wen Z M. Fluorescence spectrophotometry and its application in environmental analytical chemistry[J]. Environmental Monitoring in China, 1992, 8(2): 45−55. doi: 10.19316/j.issn.1002-6002.1992.02.018
[14] 李卿硕. 荧光光谱检测设计与研究[D]. 长春: 长春理工大学, 2008. Li Q S. Design and research of fluorescence spectroscopy detection[D]. Changchun: Changchun University of Technology, 2008.
[15] 梁锡辉, 区伟能, 任豪, 等. 激光诱导荧光检测技术[J]. 激光与光电子学进展, 2008(1): 65−72. Liang X H, Qu W N, Ren H, et al. Laser induced fluorescence detection technology[J]. Laser & Optoelectronics Progress, 2008(1): 65−72.
[16] 杨仁杰, 尚丽平, 鲍振博, 等. 激光诱导荧光快速直接检测土壤中多环芳烃污染物的可行性研究[J]. 光谱学与光谱分析, 2011, 31(8): 2148−2150. doi: 10.3964/j.issn.1000-0593(2011)08-2148-03 Yang R J, Shang L P, Bao Z B, et al. Feasibility study on laser induced fluorescence for rapid and direct detection of polycyclic aromatic hydrocarbon pollutants in soil[J]. Spectroscopy and Spectral Analysis, 2011, 31(8): 2148−2150. doi: 10.3964/j.issn.1000-0593(2011)08-2148-03
[17] 王宁. 定量测量OH基浓度的PLIF技术研究及应用[D]. 北京: 国防科学技术大学, 2009. Wang N. Research and application of PLIF technology for quantitative measurement of hydroxyl radical concentration[D]. Beijing: National University of Defense Technology, 2009.
[18] Zacharioudaki D E, Fitlis I, Kotti M. Review of fluorescence spectroscopy in environmental quality applications[J]. Molecules, 2022, 27(15): 4801. doi: 10.3390/molecules27154801
[19] Li B, Zhang D, Liu J, et al. A review of femtosecond laser-induced emission techniques for combustion and flow field diagnostics[J]. Applied Sciences, 2019, 9(9): 1906. doi: 10.3390/app9091906
[20] Radiul S M, Hazarika S. Variation of stokes shift and peak wavelength shift as a sensing probe for detection of lead in water using laser induced fluorescence resonance energy transfer[J]. Journal of Fluorescence, 2021, 31(3): 889−896. doi: 10.1007/s10895-021-02689-1
[21] 汪宝堆, 李欣悦, 张华, 等. 一种近红外二区甲基汞离子检测探针及其制备方法和应用: CN202210944448.3[P]. 2022-09-16. [22] 朱鑫琦, 张佩, 王光辉, 等. 一种激光诱导荧光的地下水重金属原位检测装置: CN202122186045.0[P]. 2022-04-01. [23] 汪宝堆, 常新月, 张华, 等. 一种超灵敏检测水中Cr(Ⅵ)离子敏感膜及其制备方法: CN202111157635.9[P]. 2021-11-19. [24] 谢胜, 杨羽娇, 吴淑军, 等. 检测汞离子的荧光探针、固态传感薄膜及其制备和应用: CN20211127665.5[P]. 2021-09-26. [25] Wan J, Zhang K, Li C, et al. A novel fluorescent chemosensor based on a rhodamine 6G derivative for the detection of Pb2+ ion[J]. Sensors and Actuators B: Chemical, 2017, 246: 696−702. doi: 10.1016/j.snb.2017.02.126
[26] Dai Y, Yao K, Fu J, et al. A novel 2-(hydroxymethyl)quinolin-8-ol-based selective and sensitive fluorescence probe for Cd2+ ion in water and living cells[J]. Sensors and Actuators B: Chemical, 2017, 251: 877−884. doi: 10.1016/j.snb.2017.05.103
[27] Pang B J, Li Q, Li C R, et al. A highly selective and sensitive coumarin derived fluorescent probe for detecting Hg2+ in 100% aqueous solutions[J]. Journal of Luminescence, 2019, 205: 446−450. doi: 10.1016/j.jlumin.2018.09.042
[28] Pan J, Li Q, Zhou D, et al. Ultrasensitive aptamer biosensor for arsenic(Ⅲ) detection based on label-free triple-helix molecular switch and fluorescence sensing platform[J]. Talanta, 2018, 189: 370−376. doi: 10.1016/j.talanta.2018.07.024
[29] Wu Y, Shi Y, Deng S, et al. Metal-induced G-quadruplex polymorphism for ratiometric and label-free detection of lead pollution in tea[J]. Food Chemistry, 2020, 343: 128425. doi: 10.1016/j.foodchem.2020.128425
[30] Su M, Liu C, Zhang Y, et al. Rational design of a water-soluble TICT-AIEE-active fluorescent probe for mercury ion detection[J]. Analytica Chimica Acta, 2022, 1230: 340337. doi: 10.1016/j.aca.2022.340337
[31] Wu S, Yang Y, Cheng Y, et al. Fluorogenic detection of mercury ion in aqueous environment using hydrogel-based AIE sensing films[J]. Aggregate, 2022, 4: e287. doi: 10.1002/agt2.287
[32] He J, Yun L, Cheng X. Organic-soluble chitosan-g-PHMA (PEMA/PBMA)-bodipy fluorescent probes and film by RAFT method for selective detection of Hg2+/Hg+ ions[J]. International Journal of Biological Macromolecules, 2023, 237: 124255. doi: 10.1016/j.ijbiomac.2023.124255
[33] Yuan H, Ren T, Luo Q, et al. Fluorescent wood with non-cytotoxicity for effective adsorption and sensitive detection of heavy metals[J]. Journal of Hazardous Materials, 2021, 416: 126166. doi: 10.1016/j.jhazmat.2021.126166
[34] Shi Y, Li W, Feng X, et al. Sensing of mercury ions in porphyra by copper@gold nanoclusters based ratiometric fluorescent aptasensor[J]. Food Chemistry, 2020, 344(5): 128694. doi: 10.1016/j.foodchem.2020.128694
[35] Yao J, He Y, Li P Y, et al. Magnified fluorescent aptasensors based on a gold nanoparticle-DNA hybrid and DNase Ⅰ for the cycling detection of mercury(Ⅱ) ions in aqueous solution[J]. Industrial & Engineering Chemistry Research, 2019, 58(47): 21201−21207. doi: 10.1021/acs.iecr.9b03622
[36] Wang Y, Lv M, Chen Z, et al. A fluorescence resonance energy transfer probe based on DNA-modified upconversion and gold nanoparticles for detection of lead ions[J]. Frontiers in Chemistry, 2020, 8: 238. DOI: 10.3389/fchem.2020.00238.eCollection 2020.
[37] Liu R, He B, Jin H, et al. A fluorescent aptasensor for Pb2+ detection based on gold nanoflowers and RecJf exonuclease-induced signal amplification[J]. Analytica Chimica Acta, 2022, 1192: 339329. doi: 10.1016/j.aca.2021.339329
[38] Yang Y, Yang Y, Xiao X, et al. One-pot synthesis of N-doped graphene quantum dots as highly sensitive fluorescent sensor for detection of mercury ions water solutions[J]. Materials Research Express, 2019, 6(9): 095615. doi: 10.1088/2053-1591/ab3006
[39] Abdolmohammad-Zadeh H, Azari Z, Pourbasheer E. Fluorescence resonance energy transfer between carbon quantum dots and silver nanoparticles: Application to mercuric ion sensing[J]. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 2020, 245: 118924. doi: 10.1016/j.saa.2020.118924
[40] Zhou J, Li B, Qi A, et al. ZnSe quantum dot based ion imprinting technology for fluorescence detecting cadmium and lead ions on a three-dimensional rotary paper-based microfluidic chip[J]. Sensors and Actuators B: Chemical, 2019, 305: 127462. doi: 10.1016/j.snb.2019.127462
[41] Bandia R, Dadigalaa R, Gangapuram B R, et al. Green synthesis of highly fluorescent nitrogen-doped carbon dots from Lantana camara berries for effective detection of lead(Ⅱ) and bioimaging[J]. Journal of Photochemistry and Photobiology B: Biology, 2018, 178: 330−338. doi: 10.1016/j.jphotobiol.2017.11.010
[42] Khoshbin Z, Moeenfard M, Zahraee H, et al. A fluorescence imaging-supported aptasensor for sensitive monitoring of cadmium pollutant in diverse samples: A critical role of metal organic frameworks[J]. Talanta, 2022, 246: 123514. doi: 10.1016/j.talanta.2022.123514
[43] Chen G, Bai W, Jin Y, et al. Fluorescence and electrochemical assay for bimodal detection of lead ions based on metal-organic framework nanosheets[J]. Talanta, 2021, 232: 122405. doi: 10.1016/j.talanta.2021.122405
[44] Hou L, Song Y, Xiao Y, et al. ZnMOF-74 responsive fluorescence sensing platform for detection of Fe3+[J]. Microchemical Journal, 2019, 150: 104154. doi: 10.1016/j.microc.2019.104154
[45] Zhang X X, Zhang W J, Li C L, et al. Eu3+-postdoped UIO-66-type metal-organic framework as a luminescent sensor for Hg2+ detection in aqueous media[J]. Inorganic Chemistry, 2019, 58(6): 3910−3915. doi: 10.1021/acs.inorgchem.8b03555
[46] Valeur B. Design principles of fluorescent molecular sensors for cation recognition[J]. Coordination Chemistry Reviews, 2000, 205: 3−40. doi: 10.1016/s0010-8545(00)00246-0
[47] Fan Y, Li J, Amin K, et al. Advances in aptamers, and application of mycotoxins detection: A review[J]. Food Research International, 2023, 170: 113022. doi: 10.1016/j.foodres.2023.113022
[48] Wu L, Wang Y, Xu X, et al. Aptamer-based detection of circulating targets for precision medicine[J]. Chemical Reviews, 2021, 121: 12035−12105. doi: 10.1021/acs.chemrev.0c01140
[49] Luo J D, Xie Z L, Lam W Y, et al. Aggregation-induced emission of 1-methyl-1, 2, 3, 4, 5-pentaphenylsilole[J]. Chemical Communications, 2001, 18: 1740−1741. doi: 10.1039/B105159H
[50] Liu B, Zhuang J Y, Wei G. Recent advances in the design of colorimetric sensors for environmental monitoring[J]. Environmental Science: Nano, 2020, 7(8): 2195−2213. doi: 10.1039/d0en00449a
[51] Lei X, Li H, Luo Y, et al. Novel fluorescent nanocellulose hydrogel based on gold nanoclusters for the effective adsorption and sensitive detection of mercury ions[J]. Journal of the Taiwan Institute of Chemical Engineers, 2021, 123: 79−86. doi: 10.1016/j.jtice.2021.05.044
[52] Ren J, Ledwaba M, Musyoka N M, et al. Structural defects in metal–organic frameworks (MOFs): Formation, detection and control towards practices of interests[J]. Coordination Chemistry Reviews, 2017, 349: 169−197. doi: 10.1016/j.ccr.2017.08.017
[53] Ghosh S, Steinke F, Rana A, et al. A fluorescent zirconium organic framework displaying rapid and nanomolar level detection of Hg(Ⅱ) and nitroantibiotics[J]. Inorganic Chemistry Frontiers, 2022, 9: 859−869. doi: 10.1039/D1QI01190A
[54] Guo X R, Wei Y, Zeng Q, et al. Fast and selective detection of mercury ions in environmental water by paper-based fluorescent sensor using boronic acid functionalized MoS2 quantum dots[J]. Journal of Hazardous Materials, 2020, 381: 120969. doi: 10.1016/j.jhazmat.2019.120969
[55] Wu F N, Zhu J, Weng G J, et al. Gold nanocluster composites: Preparation strategies, optical and catalytic properties, and applications[J]. Journal of Materials Chemistry C, 2022, 10: 14812−14833. doi: 10.1039/D2TC02095E
[56] Dai R, Zhang Y, Huang K, et al. Recent advances in the visual detection of ions and molecules based on gold and silver nanoclusters[J]. Analytical Methods, 2022, 14(29): 2820−2832. doi: 10.1039/d2ay00618a
[57] Shang L, Xu J, Nienhaus G U. Recent advances in synthesizing metal nanocluster-based nanocomposites for application in sensing, imaging and catalysis[J]. Nano Today, 2019, 28: 100767 . doi: 10.1016/j.nantod.2019.100767
[58] Yan Y, Yu H, Zhang K, et al. Dual-emissive nanohybrid of carbon dots and gold nanoclusters for sensitive determination of mercuric ions[J]. Nano Research, 2016, 9(7): 2088−2096. doi: 10.1007/s12274-016-1099-5
[59] Kirkwood N, Monchen J O V, Crisp R W, et al. Finding and fixing traps in Ⅱ–Ⅵ and Ⅲ–Ⅴ colloidal quantum Dots: The importance of Z-type ligand passivation[J]. Journal of the American Chemical Society, 2018, 140(46): 15712−15723. doi: 10.1021/jacs.8b07783
[60] Roghabadi F A, Aghmiuni K O, Ahmadi V, et al. Optical and electrical simulation of hybrid solar cell based on conjugated polymer and size-tunable CdSe quantum dots: Influence of the QDs size[J]. Organic Electronics, 2016, 34: 164−171. doi: 10.1016/j.orgel.2016.04.013
[61] Drummen G. Fluorescent probes and fluorescence (microscopy) techniques—Illuminating biological and biomedical research[J]. Molecules, 2012, 17(12): 14067−14090. doi: 10.3390/molecules171214067
[62] Zhang L, Li P, Feng L, et al. Synergetic Ag2S and ZnS quantum dots as the sensitizer and recognition probe: A visible light-driven photoelectrochemical sensor for the “signal-on” analysis of mercury(Ⅱ)[J]. Journal of Hazardous Materials, 2020, 387: 121715. doi: 10.1016/j.jhazmat.2019.121715
[63] Patir K, Gogoi S K. Facile synthesis of photoluminescent graphitic carbon nitride quantum dots for Hg2+ detection and room temperature phosphorescence[J]. ACS Sustainable Chemistry & Engineering, 2017, 6(2): 1732−1743. doi: 10.1021/acssuschemeng.7b03008
[64] Razavi S A A, Morsali A. Metal ion detection using luminescent-MOFs: Principles, strategies and roadmap[J]. Coordination Chemistry Reviews, 2020, 415: 213299. doi: 10.1016/j.ccr.2020.213299
[65] Wu T, Gao X J, Ge F, et al. Metal–organic frameworks (MOFs) as fluorescence sensors: Principles, development and prospects[J]. CrystEngComm, 2022, 24(45): 7881−7901. doi: 10.1039/d2ce01159j
[66] Wu S, Min H, Shi W, et al. Multicenter metal-organic framework-based ratiometric fluorescent sensors[J]. Advanced Materials, 2019, 32(3): 1805871. doi: 10.1002/adma.201805871
[67] Zhang D S, Gao Q, Chang Z, et al. Rational construction of highly tunable donor–acceptor materials based on a crystalline host–guest platform[J]. Advanced Materials, 2018, 30(50): 1804715. doi: 10.1002/adma.201804715
[68] Wang Q, Ke W, Lou H, et al. A novel fluorescent metal-organic framework based on porphyrin and AIE for ultra-high sensitivity and selectivity detection of Pb2+ ions in aqueous solution[J]. Dyes and Pigments, 2021, 196: 109802. doi: 10.1016/j.dyepig.2021.109802
[69] Samanta P, Desai A V, Sharma S, et al. Selective recognition of Hg2+ ion in water by a functionalized metal–organic framework (MOF) based chemodosimeter[J]. Inorganic Chemistry, 2018, 57(5): 2360−2364. doi: 10.1021/acs.inorgchem.7b02426
[70] 汪宝堆, 常新月, 张华, 等. 一种超灵敏检测水中Cr(Ⅵ)离子敏感膜及其制备方法: CN202111157635.9[P]. 2021-09-30. [71] Long X T, liu F, Zhou X, et al. Estimation of spatial distribution and health risk by arsenic and heavy metals in shallow groundwater around Dongting Lake Plain using GIS mapping[J]. Chemosphere, 2020, 269: 128698. doi: 10.1016/j.chemosphere.2020.128698
[72] Dong L, Zhang J, Guo Z, et al. Distributions and interactions of dissolved organic matter and heavy metals in shallow groundwater in Guanzhong Basin of China[J]. Environmental Research, 2022, 207: 112099. doi: 10.1016/j.envres.2021.112099




 下载:
下载:

 京公网安备 11010202008159号
京公网安备 11010202008159号