Multi-element Accurate Analysis of Sulfide Minerals by Low-temperature Ablation LA-ICP-MS
-
摘要:
硫化物矿物中元素含量及其分布可示踪硫化物成矿过程、辨别金属来源和沉积过程的物理化学条件,在地质学、矿床学等领域具有重要的应用价值。激光剥蚀电感耦合等离子体质谱(LA-ICP-MS)已成功应用于硫化物矿物元素微区分析研究,但激光与物质作用产生的热效应严重制约分析结果的可靠性。本文建立了一种高精密度、高准确度的低温剥蚀LA-ICP-MS测定硫化物矿物多元素方法。采用自行研制的Peltier低温剥蚀池可有效抑制硫化物矿物LA-ICP-MS分析中的热效应,提高分析结果的精密度和准确度。扫描电子显微镜(SEM)表明:在低温(−30℃)条件下可在一定程度地抑制激光剥蚀引起的热效应,减少样品熔化和气溶胶气相再沉积;而通过气溶胶颗粒分析发现低温剥蚀可以减小样品气溶胶颗粒的平均尺寸,得到的颗粒粒径分布范围也较小。不同元素信号强度的精密度(RSD)从常温下的20.1%~34.4%改善到11.5%~15.8%,元素的检出限为0.054~0.077μg/g。将该低温LA-ICP-MS系统应用于实验室内部标样黄铜矿Ccp-1分析,测定值与参考值之间的标准偏差在7%以内。
Abstract:BACKGROUNDMicro-geochemical information of sulfide minerals plays a crucial role in the field of geochemistry, allowing discovery of the formation mechanism and evolution process of sulfide minerals by analyzing their element composition characteristics. LA-ICP-MS is currently the most popular microanalysis technology used for sulfide analysis, having yielded successful results. Due to their unique physical and chemical properties, sulfide mineral samples show different laser ablation behavior to conventional geological samples. The most intuitive phenomenon is the melting of ablation carters caused by laser thermal effect and the deposition of a large number of material particles around the ablation carters, which is the main factor limiting the precision and accuracy of sulfide sample analysis. Walting et al[13] found that direct quantitation of multi-elements in sulfide minerals by infrared laser (1064-nm Nd:YAG laser) was impossible, which was because the strong thermal effect generated by the infrared laser will lead to severe large particle aerosol redeposition. It is reported that ablation systems with shorter wavelengths, such as ultraviolet lasers, including the 266 and 213nm laser, can be used to obtain acceptable analytical accuracy by reducing the thermal effect and aerosol particle size, but a poor precision was still observed[16-17]. Guillong et al[19] conducted a comparative study of 266, 213 and 193nm lasers and found that there were finer particle sizes of the aerosols and the weaker thermal effect when using 193nm laser ablation, and the RSDs of all elements less than 20% were obtained. In other words, collisions between photons and matter intensify in deep ultraviolet laser ablation systems (193nm) with shorter wavelength[20-21] and can help reduce the melt zone and aerosol particle size. However, there is still a slight thermal effect during 193nm UV laser ablation. Fernández et al[22] found that there is still a melting layer during 193nm laser ablation, and it leads to the formation of large particle aerosols. Different methods have been proposed to improve the thermal effect during laser ablation of sulfide. Muller et al[25] found that the precision of line scanning could be improved by 50% compared to spot ablation. Guillong’s results showed that adding a small amount of hydrogen to the analysis could increase the sensitivity of the 47 elements in the test by two to four times[26]. Moreover, research has focused on improving the thermal effect of sulfide minerals from shorter pulse width lasers and aerosol particle transport[27-31]. However, there are still some thermal effects in the process of deep ultraviolet and short wavelength laser ablation, and how to inhibit the thermal effect in the process of ablation to obtain effective analysis results is still a difficulty in the analysis of sulfide mineral elements. The LA-ICP-MS low temperature ablation cell is an ablation system developed in recent years, whose main function is to provide a low temperature ablation environment to realize the effective analysis of cells, blood and other samples. The low-temperature ablation cell may be a new approach to resolve the thermal effect during sulfide mineral ablation.
OBJECTIVESIn order to establish a high precision and high accuracy multi-element analysis method for sulfide minerals.
METHODSThe use of a designed cryogenic ablation cell suppressed the thermal effect and refined aerosol particle sizes, which improved analytical precision and accuracy significantly. To explore the mechanism of sulfide ablation at low temperature, the aerosols ablated at low temperature were collected using an aerosol collection setup consisting of a membrane with an aperture of 0.1μm, which was installed at the outlet of the ablation cell. According to the micro-analysis results, the laser ablation behavior under low temperature ablation environment was further discussed.
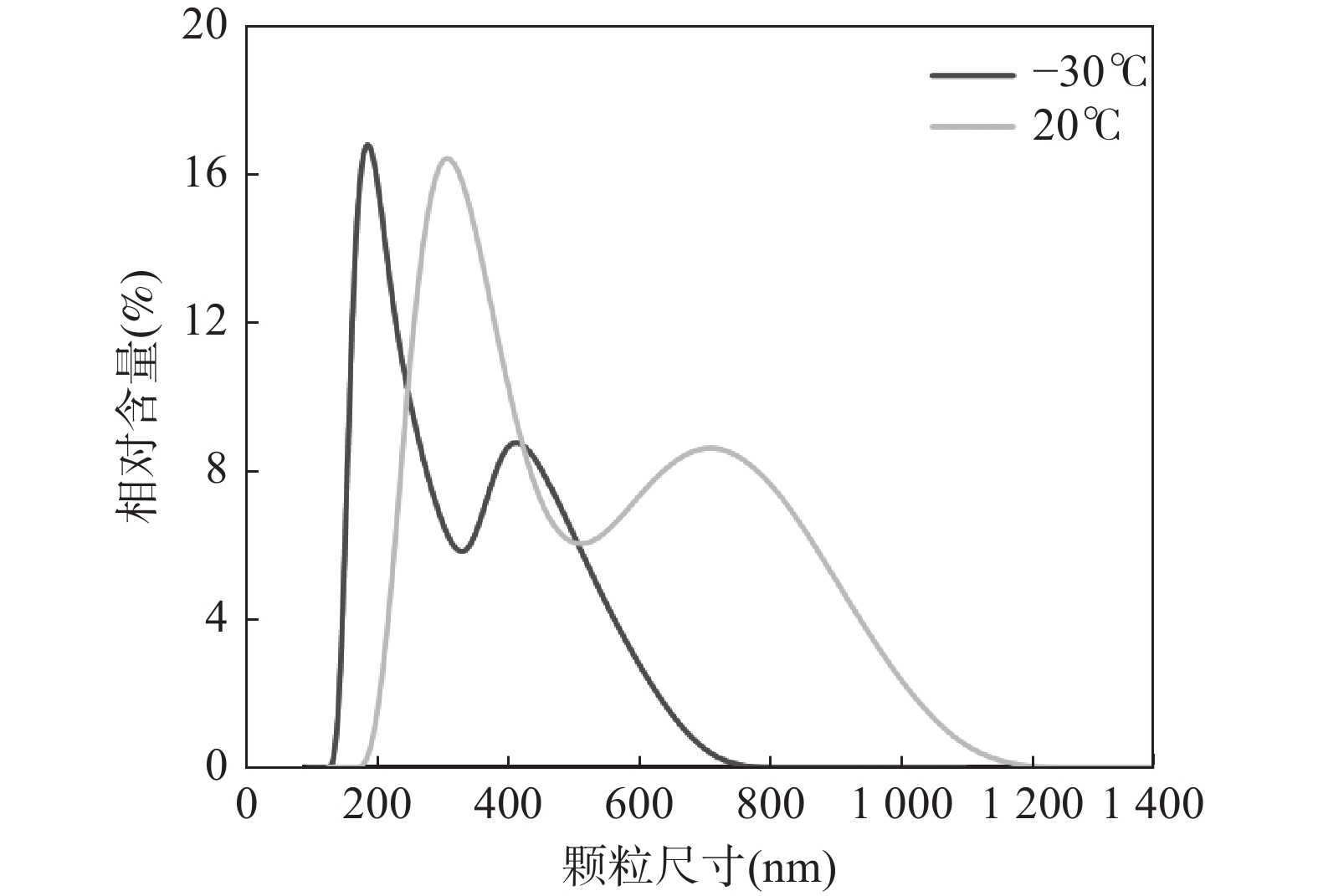
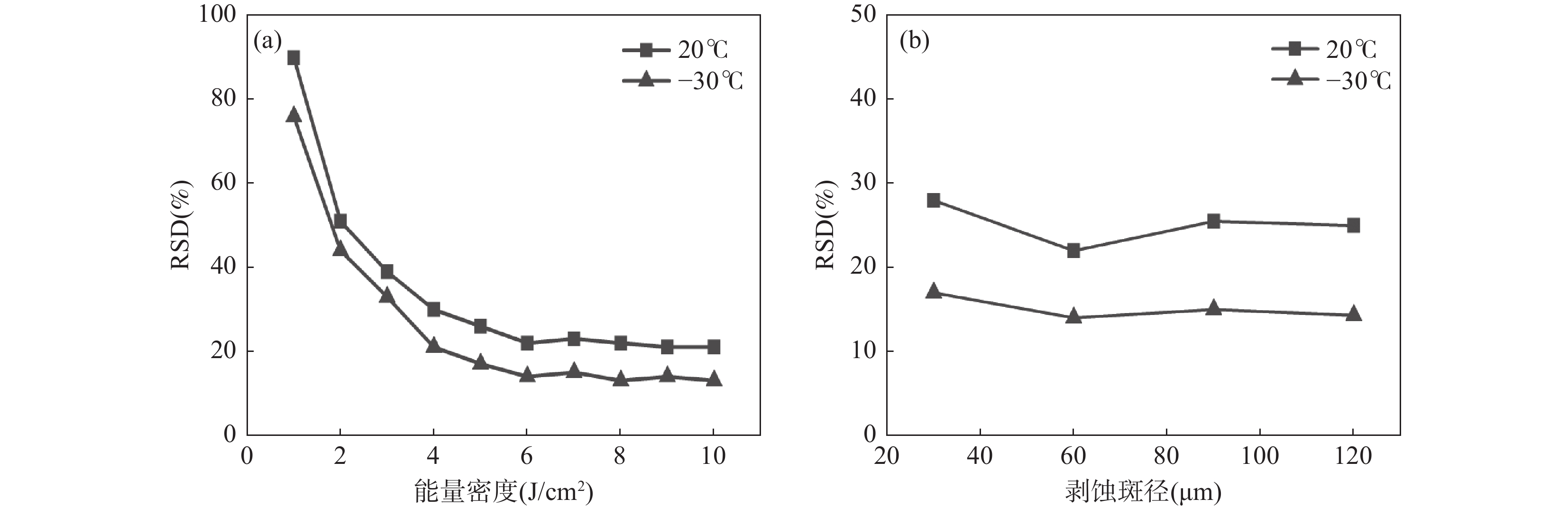
RESULTSA precision and accuracy method for multi-elements analysis of sulfide minerals using CLA-ICP-MS (laser ablation inductively coupled plasma mass spectrometry with a cryogenic ablation cell) was described. Ablation craters were investigated via scanning electron microscope (SEM) images to compare the amounts of melt produced. SEM measurements showed significant differences in melting between the low temperature (−30℃) and room temperature (20℃). The diameters and size distribution of particles were measured from nanometer particle potentiometer images of the collected ablated aerosol. Particles ablated using cryogenic ablation cell were smaller in average diameter (190nm and 400nm) and shorter in distribution range (570nm). Compared to the precision of time-resolved signal during laser ablation processes between the two temperatures, the precision was significantly improved and the RSD was reduced from 20.1%-34.4% to 11.5%-15.8% with a cryogenic ablation cell. A designed cryogenic ablation cell in sulfide sample analysis was utilized to minimize the thermal effect and improve analytical precision and signal intensity. In this study, the CRM (MASS-1) sample was analyzed with spot ablation mode at low (−30℃) and room (20℃) temperatures, respectively, and the RSDs of three times parallel analysis at these two temperatures were compared. At room temperature, the RSDs of elemental signals ranged from 20.1% to 34.4%. In contrast, the RSD of elemental signals was less than 15.8% when the sample was ablated at low temperature (Fig.2a). The significant improvements may be attributed to low ablation temperature, which suppress the thermal effect. Moreover, the signal intensities of elements improved by approximately 11% to 52% with the decrease in temperature of the cryogenic ablation cell (Fig.2b). Fig.2c shows the time-resolved signals of the MASS-1 sample at room temperature, the significant fluctuations and spikes could be observed, and the RSDs of all elemental signals was more than 20.1%. Interestingly, the signals at low temperature exhibited ideal stability, and the RSDs of elemental signals were less than 15.8%, as shown in Fig.2d. In order to explore the reasons for improving the analytical performance of low temperature, the morphology of ablation under different temperature conditions of two standard sulfide samples were discussed. SEM images of four ablation craters on chalcopyrite and pyrite were taken to investigate the effect of temperature on the ablation process (Fig.3). The sulfide samples were ablated using a 193nm excimer laser with a spot size of 60nm and a fluence of 8J/cm2. The ablation craters on the chalcopyrite showed a two-layer cyclic structure, in which the inner layer was a light-colored melting zone, and the outer layer was a white aerosol vapor sediment. At low temperature, there were fewer melt layers and thinner grain sediment zone than those at room temperature (Fig.3a, Fig.3b). However, the melting zone around the ablation craters of pyrite were more irregular. More of the unwanted ablation was melted away at room temperature (Fig.3c, Fig.3d). The craters formed at room temperature (Fig.3a, Fig.3c) showed a more serious melting phenomenon than those formed at low temperature (Fig.3b, Fig.3d), as evidenced by the abundance of molten ejecta around the former, especially at high laser energy densities. In contrast, the low temperature craters showed no obvious melting phenomenon and had a flatter bottom with a reduced number of large molten spherical particles. The use of CLA-ICP-MS weakened the melting phenomenon, thereby generating smaller aerosol particles, which further improved the aerosol transport and ionization efficiency. A particle size collection experiment was conducted to explore the distribution of aerosol particles at different temperatures. SEM images were used to analyze the shapes and sizes of particles that were collected on a membrane with an aperture of 0.1μm at room temperature (20℃) and low temperature (−30℃). The same sample chamber and 1m of tubing were used to transport the particles, and the ablation pulses continuously for 2min. The SEM images showed that the particles produced at room temperature were larger and formed large agglomerates (Fig.4a), whereas the particles produced at −30℃ were smaller and there were fewer agglomerations (Fig.4c). The shape of the agglomerates and their connection by filaments suggested strong charge during particle formation, which was more prominent at room temperature. Additionally, there were more single large particles produced at room temperature (Fig.4b), while there were fewer particles at −30℃ (Fig.4d). Comparative measurements were conducted using 193nm laser to investigate the influence of temperatures on particle size distribution. Fig.4 shows a typical size distribution for LA under He atmosphere. The left part of Fig.5 shows a distribution of aerosols produced by ablation of 2min pulses at room temperature. The peak heights of mean diameter in this distribution were determined to be approximately 300nm and 700nm, respectively. Similarly, particle size distribution at −30℃ also presented a bimodal pattern, which was consistent with previous studies. The average diameters were 190nm and 400nm, both smaller than at room temperature, while the peak width was shorter. The chemical composition of fine particles produced at low temperature is closer to the sample body, improving the transport and ionization of aerosol in ICP, reducing element fractionation, and enhancing the signal strength and stability, thereby improving the analytical performance of ICP-MS.
CONCLUSIONSA new high-precision and accuracy method for determination of trace elements in sulfide minerals has been developed using the CLA-ICP-MS system. This method reduces thermal effect and decreases particle size during the ablation process, improving precision by freezing sulfide samples with a designed cryogenic ablation cell. Low temperature results in better data because fewer large particles are produced; sedimentation around the ablation crater and during transport is reduced, while ionization efficiency in ICP is higher. The precision calculated for transient signals decreases obviously if the sample is kept at low temperature (−30℃) compared to room temperature (20℃), while the sensitivity improved slightly. The deviation of all elements between the test values and the standard values falls within 7% by CLA-ICP-MS. In future work, it will be necessary to investigate even lower temperatures, as low temperatures can increase aerosol viscosity and affect analysis results. It is also worth exploring whether the performance of a long pulse width laser can be improved by lowering the temperature to match that of a short pulse width laser.
-
X射线荧光光谱定性分析技术经过长期的应用及发展,其应用范围也越来越广泛[1-4]。目前,XRF所带的定性分析软件(SQX)可自动对扫描谱图进行搜索和匹配,包括确定峰位、背景和峰位的净强度[5-7],并从XRF特征谱线数据库中配对确定元素的谱线,这对从事XRF的分析者而言非常便利[8-10]。近年来刘岩等[11]采用XRF无标样分析法检测催化剂,测定结果的相对标准偏差小于1.3%;张红菊等[12]采用XRF无标样分析法检测轻合金铝合金中的主量元素,其测量值与认定值的相对误差低于±5%,测量结果都具有很好的可靠性和准确度。
自然界矿物种类复杂,应用XRF半定量分析软件(SQX)分析未知样品时,由于SQX软件仅对样品中9F~92U元素进行半定量分析,而对H2O、C这些参数不能直接测定。对于烧失量(LOI)、结晶水(H2O+)含量较高的铝土矿,二氧化碳含量较高的碳酸盐矿物,硫、碳含量较高的硫化物金属矿这类高烧失量矿物样品,平衡归一化计算时对未知样品中的Al2O3、SiO2、CaO、MgO、Fe等主要元素分析结果影响较大,半定量分析数据准确度较低。这就要求XRF分析人员需要掌握未知样品的来源及基本情况,根据测定结果对各元素在样品中的结构状态进行评估,选用更为合理的校正模式,提高半定量分析的准确性[13-15]。为了解决这个问题,本文提出了一种校正模式。该校正模式根据半定量分析初步结果,采用重量法、碘量法、酸碱测定法、红外光谱法有选择性地对未知样品中的LOI、S、C、H2O+等项目进行定量分析,然后将定量分析结果输入SQX该参数的固定结果中,二次平衡归一计算得出新的半定量分析结果。应用该校正模式校正后,铝土矿、碳酸盐矿物、硫化物金属矿等高烧失量矿物的半定量分析结果的准确度得到大幅度提高。
1. 实验部分
1.1 仪器与测量条件
ZSX PrimusⅣ型顺序扫描波长色散X射线荧光光谱仪(日本理学电机工业株式会社),端窗铑靶X射线管,工作电压20~60kV,工作电流2~160mA,铍窗厚度30μm,视野光栏0.5~30mm,准直器: S2/S4,探测器: PC/SC,分光晶体:RX 25/Ge/PET/LiF200[16-19]。测量元素范围9F~92U。BP-1型压样机(丹东北方科学仪器公司)。各元素具体的测量条件见表 1。
表 1 仪器测量条件Table 1. Measuring conditions of the XRF equipment分析元素 数据库 靶材 电流
(kV)电压
(mA)滤光片 衰减器 准直器 晶体 探测器 PHA 重元素 Standard Rh 50 60 OUT 1/1 S2 LiF(200) SC 100~300 重元素(1) Sta-Ni400 Rh 50 60 Ni-400 1/1 S2 LiF(200) SC 150~250 Ca-Kα Standard Rh 40 75 OUT 1/1 S4 LiF(200) PC 100~300 K-Kα Standard Rh 40 75 OUT 1/1 S2 LiF(200) PC 100~300 Cl-Kα Standard Rh 30 100 OUT 1/1 S4 Ge PC 150~300 S-Kα Standard Rh 30 100 OUT 1/1 S4 Ge PC 150~300 P-Kα Standard Rh 30 100 OUT 1/1 S4 Ge PC 150~300 Si-Kα Standard Rh 30 100 OUT 1/1 S4 PET PC 100~300 Al-Kα Standard Rh 30 100 OUT 1/1 S4 PET PC 100~250 Mg-Kα Standard Rh 30 100 OUT 1/1 S4 RX25 PC 100~250 Na-Kα Standard Rh 30 100 OUT 1/1 S4 RX25 PC 100~250 F-Kα Standard Rh 40 75 OUT 1/1 S4 RX25 PC 100~300 1.2 SQX分析模拟计算流程
XRF半定量分析可选择测定未知样品中F~U或Ti~U之间的元素,分析测试程序完成后会自动报出大于仪检出限的各元素的分析结果,这时应根据测试结果作一个初步判断是否需要进行SQX计算;如不需要,则可以直接报出测定结果;如测定结果与样品实际结构状态有较大差别,则需选用更为合适的校正模式、平衡组分或添加其他方法测试结果后进行SQX计算,以得到更为合理的测定结果。定性分析的基本流程见图 1。
1.3 样品制备及实验方法
为验证本文提出的半定量分析模式分析校正效果,选用国家标准物质铝土矿GBW(E)70036、碳酸盐矿物GBW07131、硫化物多金属矿GBW07166作为待测样品,在105℃下烘干2h,称取4.5±0.1g,倒入放置于平板模具上的PVC塑料环(外径40mm,内径35mm,高5mm)中,在30t压力下加压30s压制成型,编号,置于样品盒内,用X射线荧光光谱仪半定量分析方法进行测试[20-22]。仪器自动计算出各元素的含量。
根据XRF半定量初步分析结果,按化学标准方法YS/T 575.19—2007、GB/T 3286.8—2014、GB/T 3286.7—2014、GB/T 14353.12—2010、GB/T 8151.2—2012、SN/T 3598—2013、GB/T 2469—1996、YS/T 575.18—2007选择性分析未知样品中的烧失量(LOI)、硫(S)、碳(C)、结晶水(H2O+),计算出定量结果,备用。
将化学分析结果作为XRF半定量分析软件(SQX)中该元素的固定结果,重新进行平衡计算出新的半定量结果。
2. 结果与讨论
2.1 烧失量对铝土矿类型矿物半定量分析结果的影响
铝土矿是一种土状矿物,化学组成为Al2O3·nH2O,含水不定,多为单水或三水矿物[23-24]。由于XRF的局限性,对于H2O、C这些未定量的参数,其含量在铝土矿中较高[25],平衡归一化计算时会对Al2O3、SiO2、Fe2O3等元素的影响较大。这时可采用烧失量校正的方法,添加烧失量(LOI)作为该样品的固定值,运行半定量分析软件(SQX)重新计算出新的结果。将GBW(E)70036作为未知样品用XRF定性分析方法进行分析,各种校正模式的计算值与认定值对照结果见表 2。
表 2 铝土矿标准物质GBW(E)70036各种校正模式计算值与认定值对比Table 2. Calculated values and standard values of bauxite standard material GBW(E)70036 in various correction models分析元素 氧化物模式测试
结果(%)添加LOI校正结果
(%)H2O作平衡
校正结果(%)GBW(E)70036
认定值(%)氧化物模式测试结果
相对误差(%)LOI校正结果
相对误差(%)MgO 0.136 0.121 0.116 0.120 13.33 0.83 Al2O3 76.94 67.51 64.46 69.74 10.32 -3.20 SiO2 7.91 6.62 6.12 4.88 62.09 35.66 P2O5 0.159 0.132 0.121 0.120 32.50 10.00 SO3 0.182 0.00 0.139 0.047 - - K2O 1.07 0.880 0.810 0.710 50.70 23.94 CaO 0.258 0.212 0.195 0.180 43.33 17.78 TiO2 5.10 4.17 3.81 3.97 28.46 5.04 Fe2O3 7.42 5.97 5.35 6.09 21.84 -1.97 LOI * 13.70 △ 13.74 - - H2O * △ 18.26 - - - 注:“*”表示XRF不能直接分析该参数,无数据;“△”表示在LOI或H2O其中一项参数有测量结果时,另一项结果不参与校正计算;“-”表示未定值或未统计计算。 据表 2可知,GBW(E)70036以氧化物模式的测试结果与认定值误差较大,当添加LOI校正计算后,其多个元素的平均相对误差由32.8%降至12.3%,准确度大幅提高。此外,在确定未知样品是未经高温灼烧的情况下,还可以采用H2O作为平衡组分直接计算,其计算结果也与认定值较为相近。
2.2 二氧化碳与烧失量对碳酸盐类型矿物半定量分析结果的影响
碳酸盐矿物中CO2的占比较高, 而CO2是SQX软件未能定量参数之一,给定性分析结果带来较大误差。为提高定性分析的准确度,可对CO2或烧失量进行定量分析[26],添加烧失量或CO2定量分析结果作为该样品的固定值,运行SQX重新计算出新的结果。将GBW07131作为未知样品用XRF定性分析方法进行分析,各种校正模式的计算值与认定值对照结果见表 3。
表 3 碳酸盐标准物质GBW07131各种校正模式计算值与认定值对比Table 3. Calculated values and standard values of carbonate standard material GBW07131 in various correction models分析元素 氧化物模式
测试结果(%)CO2平衡
校正结果(%)添加LOI
校正结果(%)钙镁元素以碳酸盐
计平衡计算(%)GBW07131
认定值(%)氧化物模式测试
结果相对误差(%)LOI校正结果
相对误差(%)MgO 29.73 19.57 19.18 20.4 20.14 47.62 4.77 Al2O3 0.759 0.454 0.449 0.451 0.290 161.72 -54.83 SiO2 2.18 1.29 1.27 1.28 1.15 89.57 -10.43 P2O5 0.051 0.030 0.030 0.030 0.016 218.75 -87.50 SO3 0.442 0.256 0.00 0.254 - - - K2O 0.292 0.161 0.160 0.160 0.160 82.50 0.00 CaO 64.54 31.76 32.07 31.50 30.93 108.66 -3.69 TiO2 0.045 0.018 0.0186 0.0178 0.013 246.15 -43.08 MnO 0.038 0.015 0.016 0.011 0.012 216.67 -33.33 Fe2O3 0.435 0.169 0.176 0.167 0.170 155.88 -3.53 CO2 * 45.66 △ - - - - LOI * △ 45.67 - 45.73 - 0.13 注:“*”表示XRF不能直接分析该参数,无数据;“△”表示在LOI或CO2其中一项参数有测量结果时,另一项结果不参与校正计算;“-”表示未定值或未统计计算。 据表 3可知,GBW07131以氧化物模式测试结果较认定值误差较大。当添加LOI校正计算后,其多个元素的平均相对误差由122.4%降至27.2%,准确度大幅提高。此外,采用滴加稀盐酸确定未知样品是碳酸盐矿物的情况下,可以采用CO2作为平衡组分直接计算或者将CaO、MgO换算成为CaCO3、MgCO3计算模式重新平衡计算,其结果也与认定值较为相近。
2.3 碳硫元素对硫化物多金属矿类型矿物半定量分析结果的影响
硫化物多金属矿中的碳、硫元素含量较高,以氧化物模式对该类型样品进行半定量分析时误差较大。当采用化学法测定这类样品的烧失量时,硫化物金属矿中的硫在高温下会被空气中的氧替换,不仅会出现烧蚀减量,还会出现烧蚀增量,使得烧失量的结果是不准确的[27-28],因此不能把烧失量校作为该未知样品的固定值对测定结果进行平衡计算。这时可以采用化学法测定该未知样品中的C、S元素,作为该样品的固定值,运行半定量分析软件(SQX)重新计算出新的结果。将GBW07166作为未知样品用XRF半定量程序进行分析,各种校正模式的计算值与认定值对照结果见表 4。
表 4 硫化矿多金属矿标准物质GBW07166各种校正模式计算值与认定值对比Table 4. Calculated values and standard values of sulfide polymetallic ore standard material GBW07166 in various correction models分析元素 氧化物模式测试
结果(%)总硫、总碳固定
平衡计算(%)LOI平衡计算
(%)Sulfide模式
校正结果(%)GBW07166
认定值(%)氧化物模式测试
结果相对误差(%)总硫、总碳校正
结果相对误差(%)MgO 0.360 0.350 0.675 0.505 0.310 16.13 12.90 Al2O3 1.60 1.55 3.03 2.29 1.25 28.00 24.00 SiO2 3.34 3.50 6.26 4.86 3.78 -11.64 7.41 S 18.43 33.80 0.00 27.75 33.80 - - K2O 0.306 0.433 0.387 0.484 0.320 -4.38 35.31 CaO 2.05 2.02 2.61 3.27 1.96 4.59 3.06 Fe 18.22 28.58 27.45 30.84 29.60 -38.45 -3.45 Cu 15.50 28.00 30.72 28.42 24.20 -35.95 15.70 Zn 0.025 0.057 0.049 0.055 0.057 -56.14 0.00 C * 0.138 △ - - - - LOI * △ 27.04 - - - - 注:“*”表示XRF不能直接分析该参数,无数据;“△”表示在LOI或C其中一项参数有测量结果时,另一项结果不参与校正计算;“-”表示未定值或未统计计算。 据表 4可知,GBW07166以氧化物模式或添加LOI校正计算结果后,测试结果较认定值误差较大,当添加全硫、全碳校正计算结果后,其多个元素的平均相对误差由27.2%降至9.5%,准确度大幅提高。此外,在没有条件测定全硫、全碳元素时,选用SQX软件中Sulfide校正模式重新平衡计算,其结果也与认定值较为相近。
3. 应用实例
选取3件不同类型的未知样品,应用XRF半定量程序分析,根据XRF半定量初步分析结果,计算对照结果见表 5。未知样品1、2在添加烧失量(LOI)校正计算后半定量分析结果与化学法分析结果比较,多个元素的平均相对误差分别由46.2%降至18.0%和37.6%降至7.1%。未知样品3添加总硫、总碳校正计算结果后,其多个元素的平均相对误差由28.1%降至10%,准确度得到了明显提高。若与DZ/T 130—2006《地质矿产实验室测试质量管理规范》要求定量分析规范中误差允许限(Yc)相比较,除少部分项目能满足规范要求外,大部分项目还是达不到定量分析要求。但是如铝土矿中的Al2O3,碳酸盐矿物中的CaO、MgO,硫化物多金属矿中Fe、Zn、Cu、Pb等元素的相对误差均在5%以内,与DZ/T 130—2006要求较为接近。
表 5 某未知样品各种校正模式的计算值与化学分析值对比Table 5. Calculated values and chemical analysis values of various correction modes for the unknown sample样品编号 分析元素 氧化物模式测试
结果(%)平衡校准计算
结果(%)化学法测定值
(%)氧化物模式测试
结果相对误差(%)平衡校准计算结果
相对误差(%)允许限Yc
(%)Al2O3 86.97 76.11 78.01 11.49 -2.44 0.63 SiO2 2.94 1.82 1.31 124.43 38.93 4.17 Fe2O3 3.26 2.54 2.55 24.84 -0.46 5.11 TiO2 4.29 3.4 3.10 38.38 9.78 4.80 未知样品1 K2O 0.19 0.17 0.16 18.75 6.25 10.45 CaO 0.33 0.31 0.31 6.45 0.00 9.00 MgO 0.29 0.25 0.20 45.00 25.00 9.95 P2O5 0.28 0.22 0.14 100.00 61.37 10.76 LOI * 14.6 14.6 - - 2.58 Na2O 0.76 0.71 0.781 -2.59 -8.72 7.17 MgO 0.29 0.24 0.21 39.14 14.95 9.84 Al2O3 0.48 0.39 0.31 54.13 26.16 9.00 SiO2 1.15 0.93 0.83 38.66 12.45 7.05 P2O5 1.24 1.00 0.97 28.34 2.88 6.77 Fe2O3 1.70 1.21 1.18 44.18 2.13 6.41 未知样品2 S 4.72 △ 3.21 47.04 - 4.74 CaO 1.13 0.81 0.83 36.64 -2.40 7.05 Cr 18.49 13.09 12.89 43.46 1.53 2.75 Ni 22.81 16.18 16 42.55 1.12 2.47 Cu 14.53 10.31 10.33 40.62 -0.23 3.04 Zn 3.04 2.16 2.02 43.24 5.94 5.49 LOI * 37.00 37.00 - - - MgO 0.21 0.200 0.185 14.49 8.11 10.12 Al2O3 0.53 0.472 0.427 23.87 10.61 8.34 SiO2 2.18 1.818 1.650 32.00 10.15 5.83 P2O5 0.02 0.024 0.028 -34.29 -13.21 14.91 S 18.84 34.02 34.02 - - - K2O 0.05 0.048 0.042 15.44 13.31 13.77 未知样品3 CaO 0.15 0.127 0.137 7.72 -7.50 10.81 TiO2 0.03 0.031 0.026 30.98 20.78 15.18 Fe 4.52 6.942 6.720 -32.81 3.31 3.64 Cu 0.68 1.057 1.218 -44.37 -13.18 6.36 Zn 32.24 50.517 48.250 -33.18 4.70 1.15 Pb 1.58 2.505 2.646 -40.46 -5.33 5.05 C * 1.21 1.21 - - - 注:“*”表示XRF不能直接分析该参数,无数据;“△”表示在LOI有测量结果时,该项结果不参与校正计算;“-”表示未定值或未统计计算。 4. 结论
实验证明采用本文提出的校正模式进行校正,分析铝土矿、碳酸盐矿物和硫化物多金属矿中多元素的平均准确度提高了2.6~4.5倍,半定量分析结果准确度大幅提高。其中,铝土矿中的Al2O3,碳酸盐矿物中的CaO、MgO,硫化物矿物中Fe、Zn、Cu、Pb等主量元素的相对误差均在5%以内,与化学法分析结果较为相近。本方法可快速、较为准确地测定铝土矿、碳酸盐矿物和硫化物矿物中多元素的含量。
这种化学法与半定量分析软件相结合的半定量校正模式,不仅可用于铝土矿、碳酸盐矿物和硫化物矿物,还适用于烧失量较高的锰矿、磷矿等矿物的压片半定量分析[29-30]。对于硫化物矿物等多金属矿的定量全分析,因为这类矿物容易腐蚀铂坩埚而很少采用熔片制样XRF分析[31],通常采用化学分析法,但流程繁琐,本文研究方法可作为一种有效的矿石全分析的补充手段。
-
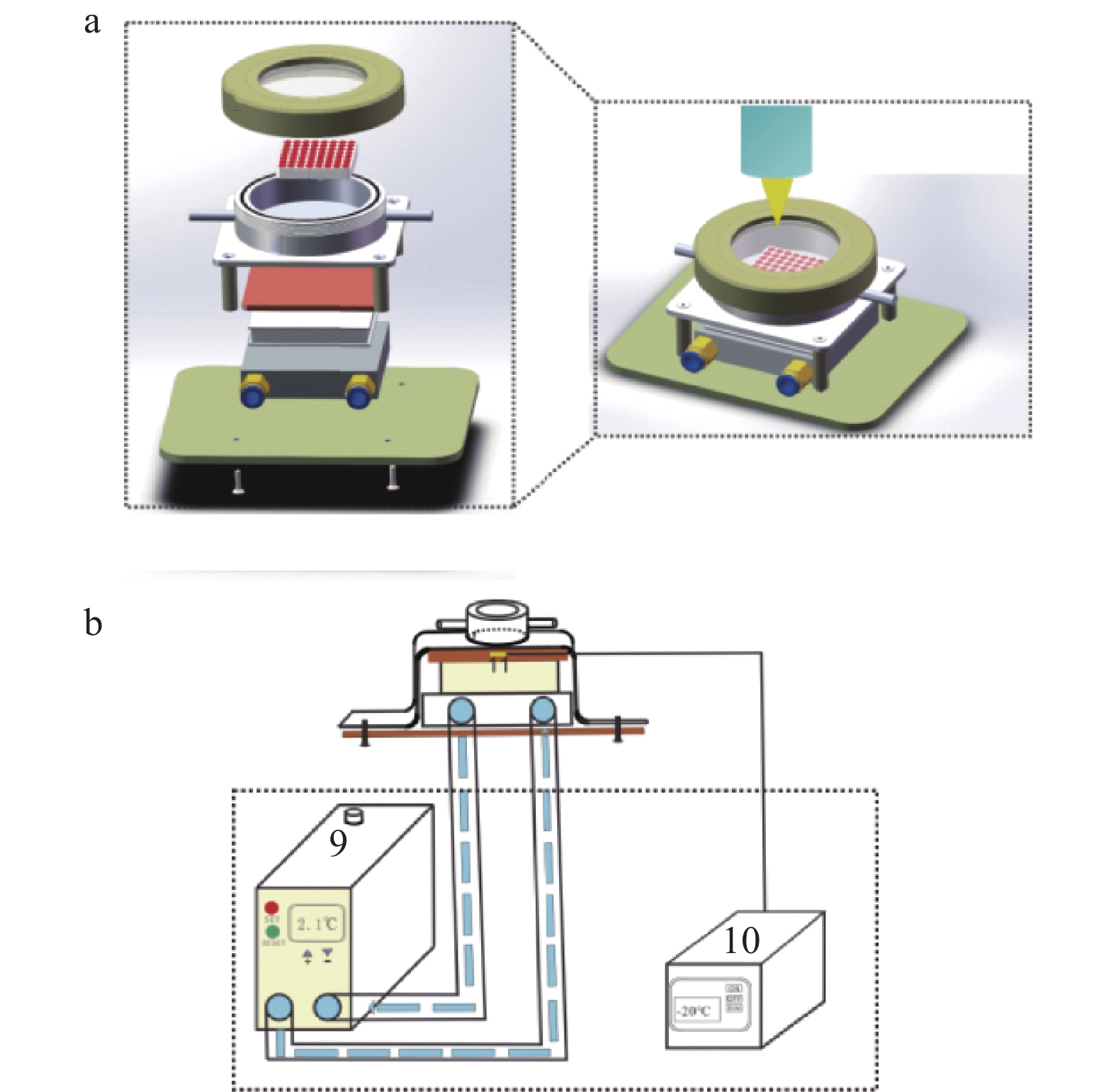
图 1 低温剥蚀池结构示意图
a. 低温剥蚀池的内部结构(1—剥蚀池;2—氟化钙窗口;3—PEEK盖;4—导热铜板;5—Peltier元件;6—水冷平台;7—底板;8—载物台);b. 剥蚀池的外部制冷系统(9—循环制冷机;10—温控系统;11—测温元件)。
Figure 1. Schematic diagram of the low-temperature ablation cell structure.
a. Interior structure of the cryogenic ablation cell (1—Ablation cell; 2—Calcium fluoride window; 3—PEEK lid; 4—Thermally conductive copper plate; 5—Peltier element; 6—Water-cooled platform; 7—Baseplate; 8—Stage); b. Ablation cell with the external refrigeration system (9—Cycle refrigeration machine; 10—Temperature control device; 11—Temperature sensor).
图 2 MASS-1在不同剥蚀温度下的剥蚀信号对比情况
a—分析信号精密度;b—分析信号强度(以室温剥蚀下元素信号强度做归一化),n=3; c—室温下元素的时间信号分辨谱图;d—低温下元素的时间信号分辨谱图。
Figure 2. Comparison of signals of MASS-1 at different ablation temperatures.
a—Analytical signal precision; b—Analytical signal intensity (Normalization is based on element signal intensity at room temperature), n=3;c—Time signal resolution spectrum of elements at room temperature; d—Time signal resolution spectrum of elements at low temperature.
图 3 硫化物矿物在不同剥蚀温度下剥蚀形貌的扫描电子显微镜图
a—20℃的黄铜矿; b—30℃的黄铜矿; c—20℃的黄铁矿; d—−30℃的黄铁矿。剥蚀条件:激光能量密度8J/cm2,剥蚀斑径为60μm。
Figure 3. Scanning electron microscopy maps of sulfide minerals morphology at different ablation temperatures.
a—Chalcopyrite at 20℃; b—Chalcopyrite at −30℃; c—Pyrite at 20℃; d—Pyrite at −30℃. Ablation conditions: laser energy density is 8J/cm2.Spot diameter is 60μm.
图 4 收集MASS-1在不同温度下激光剥蚀后气溶胶颗粒的扫描电子显微镜图
a—常温20℃下激光剥蚀颗粒,放大倍率5000×;b—常温20℃下激光剥蚀颗粒,放大倍率15000×;c—低温−30℃下激光剥蚀颗粒,放大倍率5000×;d—低温−30℃下激光剥蚀颗粒,放大倍率15000×。剥蚀条件:激光能量密度为6J/cm2,连续剥蚀2min。
Figure 4. Scanning electron microscope maps of aerosol particles after MASS-1 ablated at different temperatures.
a—Laser particles at 20℃ with 5000× magnification; b—Laser particles at 20℃ with 15000× magnification; c—Laser particles at −30℃ with 5000× magnification; d—Laser particles at −30℃ with 15000× magnification. Ablation conditions: laser energy density is 6J/cm2, continuous ablation for 2min.
表 1 LA-ICP-MS仪器工作参数
Table 1 The operating conditions of LA-ICP-MS.
电感耦合等离子体质谱
ICP-MS(7700x)激光剥蚀系统
Laser system(GeoLas HD)参数 工作条件 参数 工作条件 RF功率 1550W 激光波长 193nm 反馈功率 8W 能量密度 6J/cm2 RF电压 1.60W 剥蚀斑径 60μm 采样深度 7.5mm 激光频率 5Hz 载气(Ar)流速 0.85L/min 剥蚀气(He)流速 0.4L/min 元素 55Mn,57Fe,59Co,60Ni,63Cu,66Zn,71Ga,74Ge,75As,111Cd,208Pb 表 2 黄铜矿Ccp-1中多元素分析结果(n=3)
Table 2 The results of elemental analysis in Ccp-1 (n=3).
元素 参考值
(μg/g)测定值(−30℃ )
(μg/g)测定值(20℃ )
(μg/g)Mn 7.35±0.43 7.23±0.55 6.15±0.92 Co 5.30±0.36 5.15±0.36 4.76±0.66 Ni 7.75±0.64 7.44±0.53 6.64±0.96 Ga 8.20±0.56 8.34±0.63 7.31±1.25 Ge 8.53±1.29 8.47±0.66 6.82±1.37 As 16.51±1.19 16.96±1.32 13.47±2.56 Cd 0.24±0.01 0.26±0.03 0.21±0.05 表 3 硫化物矿物的元素分析结果(n=3)
Table 3 The analytical results of elements in sulfide samples (n=3)
元素 黄铁矿-1 黄铁矿-2 黄铁矿-3 测定值
(μg/g)SD
(μg/g)参考值
(μg/g)测定值
(μg/g)SD
(μg/g)参考值*
(μg/g)测定值
(μg/g)SD
(μg/g)参考值*
(μg/g)Mn 2.62 0.03 2.68 12.85 1.38 13.58 23.98 0.65 24.69 Co 42.04 2.53 43.76 50.07 5.91 53.1 57.25 2.68 59.33 Ni 247.2 12.11 256.36 256.04 37.01 273.86 257.58 10.29 266.36 Ga 2.95 0.18 2.89 15.76 1.23 14.66 29.61 1.44 28.72 Ge 48.05 4.65 47.25 45.97 23.14 39.04 42.03 2.5 41.04 As 13 1.92 11.26 24.77 3.36 23.49 36.52 5.72 35.01 Cd 0.09 0.01 0.09 0.55 0.03 0.56 1.05 0.09 1.07 元素 方铅矿-1 方铅矿-2 方铅矿-3 测定值
(μg/g)SD
(μg/g)参考值*
(μg/g)测定值
(μg/g)SD
(μg/g)参考值*
(μg/g)测定值
(μg/g)SD
(μg/g)参考值*
(μg/g)Mn 0.47 0.06 0.5 3.52 0.53 3.77 7.1 0.53 7.42 Co 0.37 0.05 0.41 2.7 0.21 2.82 5.58 0.26 5.78 Ni 0.52 0.06 0.55 3.29 0.52 3.72 7.89 0.84 10.92 Ga 0.64 0.06 0.63 4.93 0.35 5.15 10.59 0.53 10.81 Ge 0.57 0.06 0.48 4.39 0.12 3.62 9.65 0.47 7.33 As 0.48 0.03 0.42 3.9 0.58 3.54 7.34 0.47 8.69 Cd 0.04 0.01 0.04 0.24 0.04 0.24 0.48 0.09 0.49 元素 闪锌矿-1 闪锌矿-2 闪锌矿-3 测定值
(μg/g)SD
(μg/g)参考值*
(μg/g)测定值
(μg/g)SD
(μg/g)参考值*
(μg/g)测定值
(μg/g)SD
(μg/g)参考值*
(μg/g)Mn 2.86 0.42 3.2 12.41 0.56 12.85 25.14 3.76 26.93 Co 0.93 0.03 0.96 8.64 0.47 8.98 17.49 1.73 18.44 Ni 18.75 2.93 20.23 9.89 0.88 10.36 21.08 8.61 24.44 Ga 1.46 0.18 1.49 14.19 0.53 14.48 28.49 2.23 28.31 Ge 0.84 0.11 0.96 10.03 1.36 10.3 18.49 0.74 17.96 As 4.91 0.67 5.01 16.45 2.15 16.79 28.54 3.96 29.12 Cd 1.82 0.18 1.86 2.09 0.09 2.13 2.47 0.35 2.52 注:“*”表示硫化物实际样品的元素浓度参考值由ICP-MS测试得到。 -
[1] Cook N J, Ciobanu C L, Pring A, et al. Trace and minor elements in sphalerite: A LA-ICPMS study[J]. Geochimica et Cosmochimica Acta, 2009, 73: 4761−4791. doi: 10.1016/j.gca.2009.05.045
[2] Zhao H X, Frimmel H E, Jiang S Y, et al. LA-ICP-MS trace element analysis of pyrite from the Xiaoqinling gold district, China: Implications for ore genesis[J]. Ore Geology Reviews, 2011, 43: 142−153. doi: 10.1016/j.oregeorev.2011.07.006
[3] Deol S, Deb M, Large R R, et al. LA-ICPMS and EPMA studies of pyrite, arsenopyrite and loellingite from the Bhukia—Jagpura gold prospect, Southern Rajasthan, India: Implications for ore genesis and gold remobilization[J]. Chemical Geology, 2012, 326-327: 72−87. doi: 10.1016/j.chemgeo.2012.07.017
[4] 闫巧娟, 魏小燕, 叶美芳, 等. 激光剥蚀电感耦合等离子体质谱-电子探针分析白山堂铜矿中的黄铁矿成分[J]. 岩矿测试, 2016, 35(6): 658−666. doi: 10.15898/j.cnki.11-2131/td.2016.06.013 Yan Q J, Wei X Y, Ye M F, et al. Determination of composition of pyrite in the Baishantang copper deposit by laser ablation-inductively coupled plasma-mass spectrometry and electron microprobe[J]. Rock and Mineral Analysis, 2016, 35(6): 658−666. doi: 10.15898/j.cnki.11-2131/td.2016.06.013
[5] Ma X H, Zeng Q W, Tao S Y, et al. Mineralogical characteristics and in-situ sulfur isotopic analysis of gold-bearing sulfides from the Qilishan gold deposit in the Jiaodong Peninsula, China[J]. Journal of Earth Science, 2021, 32(1): 116−126. doi: 10.1007/s12583-020-1370-2
[6] Zhou C, Yang Z, Sun H, et al. LA-ICP-MS trace element analysis of sphalerite and pyrite from the Beishan Pb-Zn ore district, South China: Implications for ore genesis[J]. Ore Geology Reviews, 2022, 150: 105128. doi: 10.1016/j.oregeorev.2022.105128
[7] 张效瑞, 吴柏林, 雷安贵, 等. 砂岩型铀矿成矿期与非成矿期黄铁矿的微区原位Pb同位素识别特征[J]. 岩矿测试, 2022, 41(5): 717−732. Zhang X R, Wu B L, Lei A G, et al. In-situ micro-scale Pb isotope identification characteristics of metallogenic and non-metallogenic pyrites in sandstone-type uranium deposits[J]. Rock and Mineral Analysis, 2022, 41(5): 717−732.
[8] Zhang Y Y, Chu F Y, Li Z G, et al. Gold enrichment in hydrothermal sulfifides from the Okinawa Trough: An in situ LA-ICP-MS study[J]. Ore Geology Reviews, 2020, 116: 103255. doi: 10.1016/j.oregeorev.2019.103255
[9] Yang W W, Zhao H, Zhang W, et al. A simple method for the preparation of homogeneous and stable solid powder standards: Application to sulfide analysis by LA-ICP-MS[J]. Spectrochimica Acta Part B:Atomic Spectroscopy, 2021, 178: 106124. doi: 10.1016/j.sab.2021.106124
[10] Qi Y Q, Hu R Z, Gao J F, et al. Trace and minor elements in sulfides from the Lengshuikeng Ag-Pb-Zn deposit, South China: A LA-ICP-MS study[J]. Ore Geology Reviews, 2022, 141: 104663. doi: 10.1016/j.oregeorev.2021.104663
[11] Yang Q, Zhang X J, Ulrich T, et al. Trace element compositions of sulfides from Pb-Zn deposits in the Northeast Yunnan and Northwest Guizhou Provinces, SW China: Insights from LA-ICP-MS analyses of sphalerite and pyrite[J]. Ore Geology Reviews, 2022, 141: 104639. doi: 10.1016/j.oregeorev.2021.104639
[12] 员媛娇, 范成龙, 吕喜平, 等. 电子探针和LA-ICP-MS技术研究内蒙古浩尧尔忽洞金矿床毒砂矿物学特征[J]. 岩矿测试, 2022, 41(2): 211−225. doi: 10.3969/j.issn.0254-5357.2022.2.ykcs202202007 Yuan Y J, Fan C L, Lyu X P, et al. Application of EPMA and LA-ICP-MS to study mineralogy of arsenopyrite from the Haoyaoerhudong gold deposit[J]. Rock and Mineral Analysis, 2022, 41(2): 211−225. doi: 10.3969/j.issn.0254-5357.2022.2.ykcs202202007
[13] Watling R J, Herbert H K, Abell I D. The application of laser ablation-inductively coupled plasma-mass spectrometry (LA-ICP-MS) to the analysis of selected sulphide minerals[J]. Chemical Geology, 1995, 124: 67−81. doi: 10.1016/0009-2541(95)00025-H
[14] Watling R J. In-line mass transport measurement cell for improving quantification in sulfide mineral analysis using laser ablation inductively coupled plasma mass spectrometry[J]. Journal of Analytical Atomic Spectrometry, 1998, 13: 927−934. doi: 10.1039/a800337h
[15] 吴石头, 许春雪, 肖益林, 等. 193nm ArF准分子激光系统对LA-ICP-MS分析中不同基体的剥蚀行为和剥蚀速率探究[J]. 岩矿测试, 2017, 36(5): 451−459. Wu S T, Xu C X, Xiao Y L, et al. Study on ablation behaviors and ablation rates of a 193nm ArF excimer laser system for selected substrates in LA-ICP-MS analysis[J]. Rock and Mineral Analysis, 2017, 36(5): 451−459.
[16] Bacon J R, Crain J S, Vaeck L V, et al. Atomic mass spectrometry[J]. Journal of Analytical Atomic Spectrometry, 1999, 14: 1633−1659. doi: 10.1039/a905419g
[17] Günther D, Horn I, Hattendorf B. Recent trends and developments in laser ablation ICP mass spectrometry[J]. Fresenius Journal of Analytical Chemistry, 2000, 368: 4−14. doi: 10.1007/s002160000495
[18] Hergenröder R. Laser-generated aerosols in laser ablation for inductively coupled plasma spectrometry[J]. Spectrochimica Acta Part B: Atomic Spectroscopy, 2006, 61: 284−300. doi: 10.1016/j.sab.2006.02.001
[19] Guillong M, Horn I, Günther D. A comparison of 266nm, 213nm and 193nm produced from a single solid state Nd: YAG laser for laser ablation ICP-MS[J]. Journal of Analytical Atomic Spectrometry, 2003, 18: 1224−1230. doi: 10.1039/B305434A
[20] Günther D, Heinrich C A. Comparison of the ablation behaviour of 266nm Nd: YAG and 193nm ArF excimer lasers for LA-ICP-MS analysis[J]. Journal of Analytical Atomic Spectrometry, 1999, 14: 1369−1374. doi: 10.1039/A901649J
[21] Liu Y S, Hu Z C, Li M, et al. Applications of LA-ICP-MS in the elemental analyses of geological samples[J]. Chinese Science Bulletin, 2013, 58: 3863−3878. doi: 10.1007/s11434-013-5901-4
[22] Fernández B, Claverie F, Pécheyran C, et al. Direct analysis of solid samples by fs-LA-ICP-MS[J]. Trends in Analytical Chemistry, 2007, 26: 951−966. doi: 10.1016/j.trac.2007.08.008
[23] 柯于球, 张路远, 柴辛娜, 等. 硫化物矿物 LA-ICP-MS 激光剥蚀元素信号响应[J]. 高等学校化学学报, 2012, 33(2): 257−262. doi: 10.3969/j.issn.0251-0790.2012.02.008 Ke Y Q, Zhang L Y, Chai X N, et al. Elemental signal response of sulfide minerals in LA-ICP-MS microanalysis[J]. Chemical Journal of Chinese Universities, 2012, 33(2): 257−262. doi: 10.3969/j.issn.0251-0790.2012.02.008
[24] Kuhn H R, Günther D. Laser ablation-ICP-MS: Particle size dependent elemental composition studies on fifilter-collected and online measured aerosols from glass[J]. Journal of Analytical Atomic Spectrometry, 2004, 19: 1158−1164. doi: 10.1039/B404729J
[25] Mueller W, Shelley J, Rasmussen S. Direct chemical analysis of frozen ice cores by UV-laser ablation ICPMS[J]. Journal of Analytical Atomic Spectrometry, 2011, 26: 2391−2395. doi: 10.1039/c1ja10242g
[26] Guillong M, Heinrich C A. Sensitivity enhancement in laser ablation ICP-MS using small amounts of hydrogen in the carrier gas[J]. Journal of Analytical Atomic Spectrometry, 2007, 22: 1488−1494. doi: 10.1039/b709489b
[27] Bogaerts A, Chen Z, Gijbels R, et al. Laser ablation for analytical sampling: What can we learn from modeling[J]. Spectrochimica Acta Part B: Atomic Spectroscopy, 2003, 58: 1867−1893. doi: 10.1016/j.sab.2003.08.004
[28] Poitrasson F, Mao X L, Mao S S, et al. Comparison of ultraviolet femtosecond and nanosecond laser ablation inductively coupled plasma mass spectrometry analysis in glass, monazite, and zircon[J]. Analytical Chemistry, 2003, 75: 6184−6190. doi: 10.1021/ac034680a
[29] Liu C, Mao X L, Mao S S, et al. Nanosecond and femtosecond laser ablation of brass: Particulate and ICPMS measurements[J]. Analytical Chemistry, 2004, 76: 379−383. doi: 10.1021/ac035040a
[30] Telouk P, Rose-Koga E F, Albarede F. Preliminary results from a new 157nm laser ablation ICP-MS instrument: New opportunities in the analysis of solid samples[J]. Geostandards and Geoanalytical Research, 2003, 27: 5−11. doi: 10.1111/j.1751-908X.2003.tb00708.x
[31] Wohlgemuth-Ueberwasser C C, Jochum K P. Capability of fs-LA-ICP-MS for sulfide analysis in comparison to ns-LA-ICP-MS: Reduction of laser induced matrix effects[J]. Journal of Analytical Atomic Spectrometry, 2015, 30: 2469−2480. doi: 10.1039/C5JA00251F
[32] Reinhardt H, Kriews M, Miller H, et al. Laser ablation inductively coupled plasma mass spectrometry: A new tool for trace element analysis in ice cores[J]. Fresenius Journal of Analytical Chemistry, 2001, 370(5): 629−636. doi: 10.1007/s002160100853
[33] Feldmann J, Kindness A, Ek P. Laser ablation of soft tissue using a cryogenically cooled ablation cell[J]. Journal of Analytical Atomic Spectrometry, 2002, 17(8): 813−818. doi: 10.1039/b201960d
[34] Wang Y, Wei X, Liu J H, et al. Cryogenic laser ablation in a rapid cooling chamber ensures excellent elemental imaging in fresh biological tissues[J]. Analytical Chemistry, 2022, 94(23): 8547−8553. doi: 10.1021/acs.analchem.2c01736
[35] Li F, Lei X Q, Li H L, et al. Direct multi-elemental analysis of whole blood samples by LA-ICP-MS employing a cryogenic ablation cell[J]. Journal of Analytical Atomic Spectrometry, 2023, 38: 90−96. doi: 10.1039/D2JA00282E
[36] Li F, Cui H, Zhang D W, et al. Direct multi-elemental analysis of cerebrospinal fuid samples by LA−ICP−MS employing an aerosol local extraction cryogenic ablation cell[J]. Journal of Analytical and Bioanalytical Chemistry, 2023, 415: 6051−6061. doi: 10.1007/s00216-023-04878-2
[37] Wilson S A, Ridley W I, Koenig A E. Development of sulfide calibration standards for the laser ablation inductively-coupled plasma mass spectrometry technique[J]. Journal of Analytical Atomic Spectrometry, 2002, 17: 406−409. doi: 10.1039/B108787H
[38] Jarošová M, Walaszek D, Wagner B, et al. Influence of temperature on laser ablation fractionation during ICP-MS analysis for 213nm and 266nm laser wavelength micro-sampling[J]. Journal of Analytical Atomic Spectrometry, 2016, 31: 2089−2093. doi: 10.1039/C6JA00182C
[39] Koch J, von Bohlen A, Hergenröder R, et al. Particle size distributions and compositions of aerosols produced by near-IR femto- and nanosecond laser ablation of brass[J]. Journal of Analytical Atomic Spectrometry, 2004, 19: 267−272. doi: 10.1039/B310512A
[40] Li Z, Hu Z C, Günther D, et al. Ablation characteristic of ilmenite using UV nanosecond and femtosecond lasers: Implications for non-matrix-matched quantification[J]. Geostand Geoanalytical Research, 2016, 40: 477−491. doi: 10.1111/ggr.12117
[41] Longerich H P, Günther D, Jackson S E. Elemental fractionation in laser ablation inductively coupled plasma mass spectrometry[J]. Fresenius Journal of Analytical Chemistry, 1996, 355: 538−542. doi: 10.1007/s0021663550538
-
期刊类型引用(5)
1. 刘畅,胡骏翔,苑芷茜. 应用PXRF与ICP-OES法测定土壤中锰的比对研究. 环境科学与管理. 2023(07): 115-119 .  百度学术
百度学术
2. 田戈,刘卫,郭颖超,马春红,张文宇,谷周雷. 恒温振荡浸提-电感耦合等离子体发射光谱法测定土壤中的交换性锰. 中国土壤与肥料. 2023(08): 243-248 .  百度学术
百度学术
3. 杨晓红,陈丽琼,刘婉秋. X射线荧光光谱法在环境监测中的发展与应用. 理化检验-化学分册. 2022(07): 861-868 .  百度学术
百度学术
4. 徐冬梅,陈晋,张敏. WDXRF法测定污染土壤和沉积物中金属元素. 环境监测管理与技术. 2022(06): 52-55+68 .  百度学术
百度学术
5. 刘宏,付淑惠,李海霞,靳晧琛,余恒. X射线荧光光谱仪滤光片故障解决思路探讨. 四川环境. 2022(06): 254-259 .  百度学术
百度学术
其他类型引用(2)



 下载:
下载:




 京公网安备 11010202008159号
京公网安备 11010202008159号